Finding oxygen in an exoplanet's atmosphere is a clue that life may be at work. On Earth, photosynthetic organisms absorb carbon dioxide, sunlight, and water and produce sugars and starches for energy. Oxygen is the byproduct of that process, so if we can detect oxygen elsewhere, it'll generate excitement.
But researchers have also put pressure on the idea that oxygen in an exoplanet's atmosphere indicates life. It's only evidence of life if we can rule out other pathways that created the oxygen.
But scientists can't rule them out.
Earth is saturated with oxygen. It makes up 46 percent of the crust and about the same percentage of the mantle, and the atmosphere is about 20 percent oxygen.
Oxygen's presence stems from the Great Oxygenation Event (GOE) about two billion years ago. Ancient cyanobacteria evolved pigments that absorb sunlight and use it in photosynthesis. Oxygen is photosynthesis' waste product, and life has had a couple of billion years to build oxygen up in the atmosphere, mantle, and crust.
So if scientists find oxygen in an exoplanet's atmosphere, it strongly indicates that life might be at work. Simple life may be bubbling away in the planet's oceans, taking in sunlight and spewing out oxygen.
But new research has identified a source of oxygen that doesn't rely on life.
The research article is "Abiotic molecular oxygen production—Ionic pathway from sulfur dioxide," published in Science Advances. The lead author is Måns Wallner, a doctoral student in physics at the University of Gothenburg in Sweden.
The researchers have found an abiotic source of oxygen that stems from sulphur dioxide. Sulphur is not rare in celestial bodies, and since volcanoes produce sulphur and pump it into the atmosphere, terrestrial volcanic exoplanets may have oxygen in their atmospheres. And life needn't be involved.
Instead, high-energy radiation from a star can ionize the sulphur dioxide molecule. Sulphur dioxide's formula is SO2, and when it gets ionized, the molecule rearranges itself. It becomes a "double positively-charged system." Then it has a linear form with both oxygen atoms adjacent to one another and the sulphur at the other end. This is called roaming, as the oxygen atoms are free to drift around in chaotic orbits until settling into new compounds.
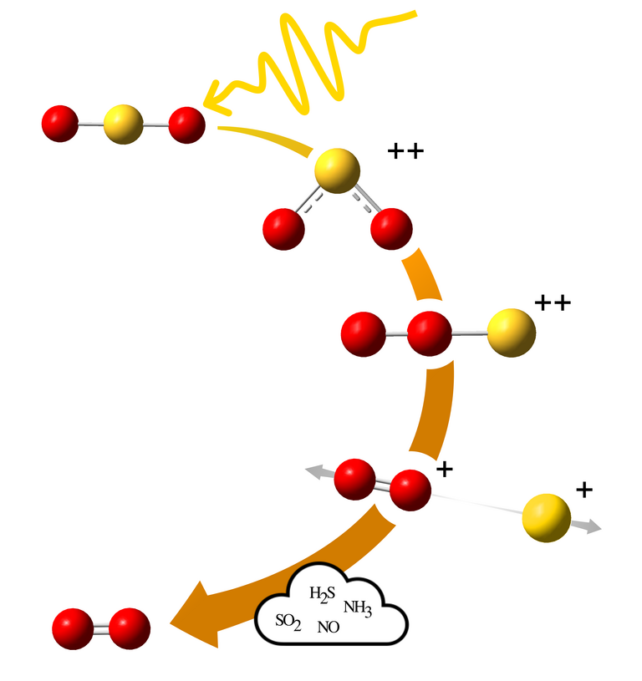
"Upon double ionization, two of the bound electrons in the molecule get ejected and can lead to changes in the angle between the atoms in the molecule," lead author Wallner said in a press release.
"Alternatively, as crucial in the present case, roaming can occur, that is, the atoms switch places, and the molecule takes on a whole new shape."
But the constituents of the molecule might not reform into SO2 again. Instead, the sulphur may break up, and a simple positively-charged oxygen molecule can remain. Then the positive charge can be neutralized by attracting an electron from another molecule. Molecular oxygen (O2) remains, and it's vital to life on Earth.
This pathway to oxygen may explain some of the oxygen we find elsewhere. Io, Ganymede, and Europa all have oxygen in their atmospheres, and roaming could be the cause.
Io is a volcanic place – the most volcanic world in the Solar System – so life is ruled out there. Ganymede and Europa have subsurface oceans, so they could potentially harbour life. But that life can't build an oxygen atmosphere like Earth life. Another explanation is required to account for the oxygen found on these moons.
This oxygen pathway may happen on Earth, too, according to the researchers.
"We also suggest in our article that this happens naturally on Earth," said Raimund Feifel, co-author of the article reporting the findings.
This ionic oxygen-forming pathway may work for other molecules, too, and that's next for the researchers. They want to know if other molecules like carbon diselenide are subjected to double ionization.
"We want to see if it also happens then, or if it was just a happy coincidence with sulphur dioxide," said Feifel.
Other researchers have tackled abiotic O2 sources. A 2014 paper presented evidence for molecular oxygen produced from CO2 when exposed to high-energy UV light.
In a 2015 paper, Japanese researchers showed that near-Ultraviolet light could produce O2 on exoplanets when interacting with water using Titania (titanium dioxide) as a catalyst.
These findings help explain how Earth had a small amount of oxygen in its atmosphere before the GOE. Since oxygen is so reactive, there must have been a replenishing source, and these pathways could be responsible.
The James Webb Space Telescope forms part of the backdrop for this research. Studying exoplanet atmospheres is one of the telescope's science objectives, and with its powerful infrared instruments, it's poised to reveal the chemical makeup of exoplanet atmospheres.
If it finds oxygen, there'll be some excitement. But as this research shows us, there's more to oxygen than life.
This article was originally published by Universe Today. Read the original article.
