Six seconds is how long it takes for Pseudomonas aeruginosa - a bacterium that's been labelled a 'superbug' - to go from a fully functional cell, to an exploded mess.
Although this sounds pretty alarming, the newly discovered ability can actually help scientists understand more about bacterial superbugs, and could eventually allow them to control antibiotic resistant infections with all-new drugs.
This new research, a collaboration between Australia, Japan and Switzerland, tips our understanding of how bacterial cells die on its head. When our human cells die, they implode, committing programmed cell death and ending up in a nice little ball that gets eaten by our immune system.
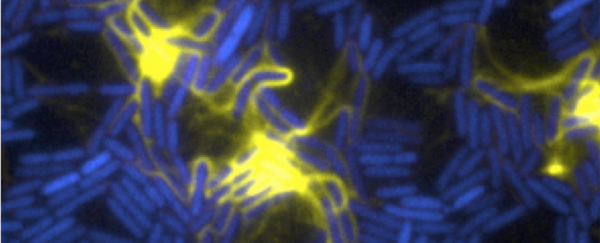
It's simple, but stops damaging cell juice leaking everywhere. And we assumed bacterial cells did the same, or worst case, just slowly leaked out when they died, leaving a slow sticky mess. But it turns out that instead of a slow burn, a few of the P. aeruginosa bacteria in the larger colony go out with a bang and explode instead. Their leftovers is what helps the remaining bacteria glue themselves together as a drug-resistant biofilm.
"When most people think about bacterial cell death, they think of the cell dying and their contents slowly leaking out, similar to what you would see with a piece of fruit rotting," said lead investigator, Cynthia Whitchurch who worked with Lynne Turnbull on the project, both from the University of Technology Sydney (UTS) in Australia.
"What's so amazing about this discovery is that we now know the bacteria have a process that enables them to actively explode, and therefore efficiently release all of their internal contents, making these available for use by the remaining members of their community."
Yeah, it's pretty unsophisticated, but it's one of the reasons why bacteria can do so much damage to us.
Many of the worst bacterial infections caused by superbugs are made even more deadly due to biofilm formation, where bacteria pile on top of each other on a surface. Here they act as a living barrier, stopping antibiotics and other antimicrobials from reaching the inside of the pile. This can make infections significantly harder to destroy, even when using the correct antibiotics.
The new research has shown that when a bacterial cell explodes, the protein and DNA are used as a kind of 'glue' for the biofilm, as well as a food source. They also add extra virulence factors to the pile of remaining bacteria, making them more dangerous to humans.
"The normal bacteria look like little rods or pills," says Whitchurch. "One day, as we looked under the microscope, we saw one of the cells turn from a hard, structured rod into a round, soft ball. Within a few more seconds, it then violently exploded - it's amazing how quickly it happens and is likely the reason it hasn't been observed before."
P. aeruginosa is a big problem in hospitals, where people with damaged immune systems, such as the elderly, surgery patients, or those with cancer, often end up infected. P. aeruginosa is also multi-drug resistant, adding to the growing list of bacterial species which can only be treated by a dwindling number of antibiotics.
Because it's so good at forming biofilms, it makes the infection even harder to clear even if the drugs do work, causing roughly 400 deaths a year in the US alone.
But it's not all bad news. The UTS researchers discovered that when a particular gene in P. aeruginosa was removed, it not only stopped the cells from exploding, but also prevented the formation of biofilms.
The team is now ready to investigate how we can trick this gene into working for us, and hopefully figure out how to stop superbugs in their tracks.
"We think there will be a two pronged approach: one is to prevent the biofilms being produced by stopping bacterial explosions, but if we can't prevent it, then the second approach will be to induce the process that cause the bacteria to explode, so they all die en masse causing the infection to clear," says Whitchurch.
Although we're not sure we want a whole bunch of bacteria exploding inside us in one go, we are looking forward to where this research can take us. Anything to stop the increasing number of superbugs, especially if it doesn't involve our last line of antibiotics.
The research was published in Nature Communications.
UTS Science is a sponsor of ScienceAlert. Find out more about their research.