Long COVID has more than 200 potential symptoms and can affect almost every organ in the body.
With more than 65 million people now estimated to live with this often-disabling condition worldwide, and numbers growing daily, there is a desperate need to understand the underlying biology driving it.
There are currently no effective treatments and no tests.
To find out more about this long COVID infection – or post-acute sequelae SARS-CoV-2 (PASC), as it is increasingly being called – researchers at the US National Institute of Health did extensive testing on 12 people with long COVID and compared those results against a group of healthy volunteers who hadn't contracted COVID.
The researchers looked for abnormalities in blood samples, cerebrospinal fluid, and MRI scans, and they ran a series of tests to detect autonomic nervous system dysfunction.
The long COVID group was mostly middle-aged women who had mild SARS-CoV-2 infections around nine months ago and were now experiencing fatigue and cognitive difficulties that severely impacted their daily life.
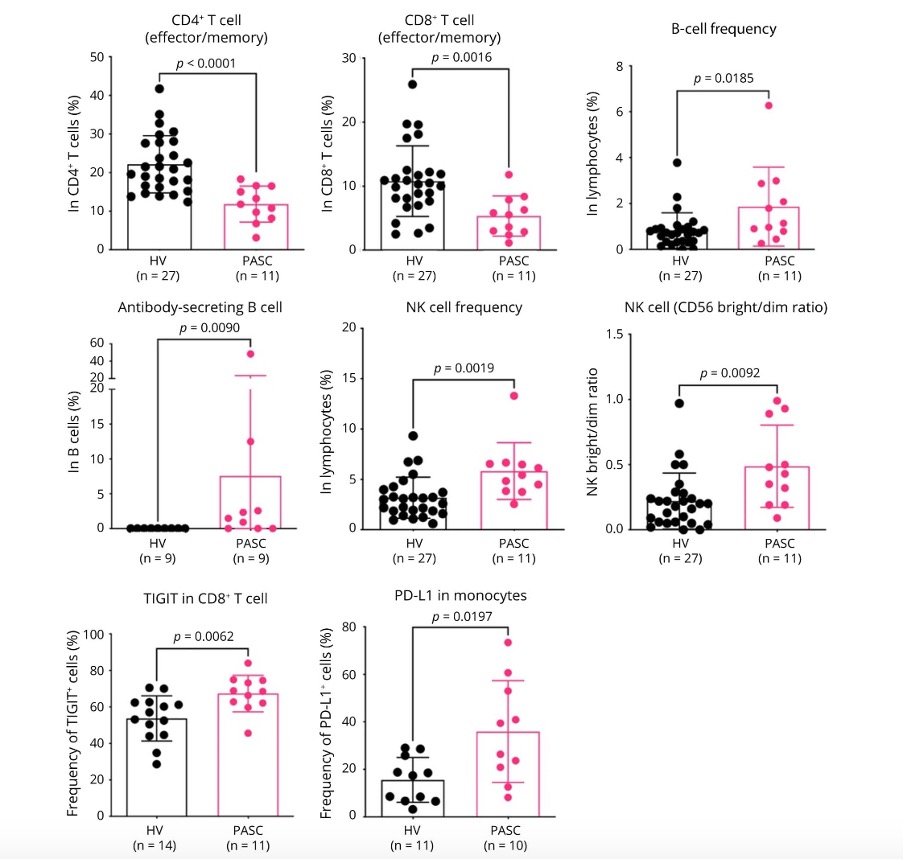
One of the most apparent disparities between people with long COVID and healthy volunteers was the immune cell count.
Compared with the control group, participants with long COVID had decreased numbers of immune cells called memory T cells; these cells usually persist for an extended period after infections, retaining the ability to recognize a specific threat and call the rest of the immune system rapidly to arms upon re-exposure.
COVID long haulers also had increased numbers of immune cells called B cells, antibody-secreting B cells, and activated natural killer cells, which detect and destroy damaged cells.
For those with long COVID, there was also an increase in immune checkpoint molecules like TIGIT and PD-L1 on immune cells, "suggesting the possibility of immune exhaustion", the researchers wrote.
"The persistence of these immune abnormalities several months after a mild infection suggests the possibility of either a persistent infection or an aberrant immune response to the infection," the authors wrote.
The researchers also examined dysfunction in the autonomic nervous system, which controls heart rate, blood pressure, and breathing.
People with long COVID often report a racing heart, dizziness, and feeling faint when moving from a sitting to a standing position, known as postural orthostatic tachycardia syndrome (POTS).
To measure these effects, researchers monitored changes to heart rate and blood pressure during the tilt-table test, where a person is moved from a lying to a standing position, and during the Valsalva maneuver, where a person blows out air while keeping their nose and mouth shut, which usually slows the heart rate down.
They found that people with long COVID were less able to control their heart rate and blood pressure when these physiological pressures were applied.
The researchers noted that the skin temperature was also lower in people with long COVID throughout the tilt-table test, suggesting vasoconstriction, where the blood vessels constrict and decrease blood flow to the skin.
While previous studies have suggested that long COVID's neurological symptoms could be due to inflammation in the brain stem and in the brain's olfactory bulb, which receives sensory inputs from the nose, the MRI did not identify any structural abnormalities in either region.
More research is needed to make head or tail of all this, but this research does suggest that immunotherapies could be explored as a treatment for long COVID in clinical trials.
"The preliminary findings […] call for further investigation and for evaluation of potential immunomodulatory agents in an effort to decrease the huge public health burden of this syndrome," the researchers write.
This paper was published in Neurology: Neuroimmunology & Neuroinflammation.
