While human beings have some impressive self-healing capabilities, we're nowhere near the level of salamanders or axolotls that can resprout whole appendages – but might that change in the future?
A team led by researchers from Texas A&M University have successfully coaxed a regenerative tissue response in mice that had a toe removed. The regrowth was "imperfect", the researchers report, but the results are definitely encouraging.
The technique used here hasn't been tested in humans, but considering the biological similarities between mammals, it's tempting to think what our own cells could do, given the right instructions.
Key to the process was the application of two proteins that send out specific signals: the first protein generates the raw material for regeneration, and a second builds tissue with it.
"This is really a two-step process," says Ken Muneoka, a regenerative biologist at Texas A&M University.
"You first shift the cells away from scarring, and then you provide the signals that tell them what to build."
That scarring is the default reaction when we get injured. Fibroblast cells are dispatched to the wound site to patch it up with scar tissue – an effective way to stop the bleeding, but in the case of a lost limb, it's not coming back.
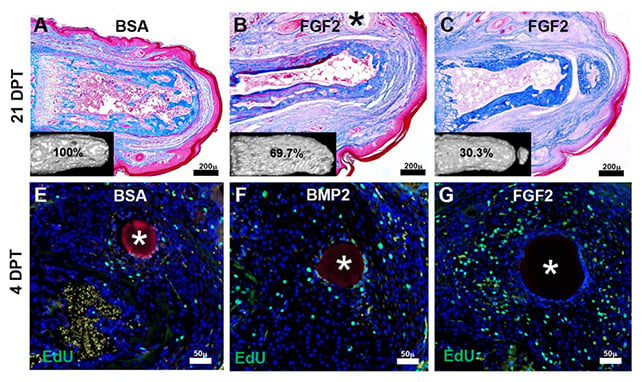
These fibroblasts are in an active, receptive state while they do their work, which is where the first signaling protein comes in: fibroblast growth factor 2 (FGF2). It essentially reprograms these cells, ready to transform into something else.
That something else is a blastema. These are temporary cell 'buds' used by animals like salamanders to prepare tissue to regrow – not just to repair the wound site, but to replace the lost appendage.
Next, bone morphogenetic protein 2 (BMP2) was applied, delivering the messages to the blastema to start building, on top of the preparation work done by FGF2.
The double protein treatment was enough to bring back bones, tendons, ligaments, and joint structures – so the skeletal and connective tissue elements of the missing toe – across dozens of attempts in tests on mice.
While the replacement digits were sometimes misshapen or too small, all the essential parts were there.
What's interesting about this approach is that it's unusual in the broader field of regenerative medicine. The focus is usually on a fresh application of stem cells, which can subsequently transform into a host of different cell types.
"You don't have to actually get stem cells and put them back in," says Muneoka of his team's approach.
"They're already there – you just need to learn how to get them to behave the way you want."
The new study builds on earlier research from the same lab, which used a similar protein signaling approach.
However, no FGF2 was used in those earlier experiments, no blastema was formed, and only part of the missing limb was regrown.
"This changes the way we think about what's possible," explains co-author and veterinary physiologist Larry Suva.
"Once you show that regeneration can be activated, it opens the door to asking entirely new questions."
It's definitely not the first time that scientists have suspected our own regenerative powers could be locked away inside us, rather than entirely absent.
Of course, if they can be found and enhanced, the implications for medical treatments are huge.
There's a lot of work to do before this latest approach can be trialed in humans, including a closer analysis of the regrowth mechanisms and producing limbs that more closely match the ones that have been lost.
Related: 'Phantom Limbs' May Not Be What We Thought, Surprising Study Finds
With BMP2 already approved for use in reconstructive surgery and FGF2 on its way to the same status, there could be more immediate benefits too: improving wound repair and reducing scarring, even if there's no actual regeneration.
"Why some animals can regenerate and others, particularly humans, can't is a big question that has been asked since Aristotle," says Muneoka.
"I've spent my career trying to understand that."
The research has been published in Nature Communications.