It took nature decades of photosynthesis, followed by eons of intense heat and pressure from geological activity to bake atmospheric carbon dioxide into the long chains of hydrocarbon that make up fossil fuels.
We don't have the luxury of millions of years to mop up the excess carbon from our atmosphere, but advances in chemistry could help us get part of the way there in a relative blink of the eye.
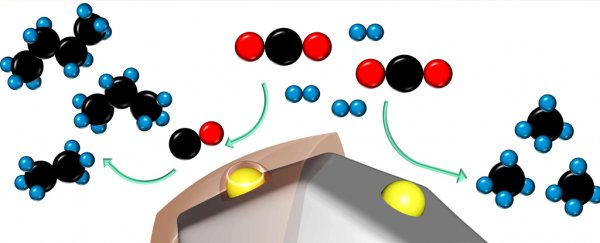
The latest advance is a new catalyst capable of churning out short-chained carbon molecules made from carbon dioxide at a rate that blows previous methods clear out of the water.
Developed by chemical engineers at Stanford University, the technology turns waste CO2 and a good dose of hydrogen into chains of ethane, propane, and even butane, all molecules that can serve as a fuel source.
"We can create gasoline, basically," says Matteo Cargnello, a chemical engineer at Stanford University.
In recent years, there's no shortage of efforts to find economical ways to pull carbon out of the air and turn it into something people will want to pay for, such as fuel or synthetic materials like plastic.
The challenge is to make something that will make a dent in the ridiculous amounts of carbon we pump into the atmosphere each and every year. That means a rapid process that can be scaled up cheaply, locking away as much carbon as possible into every molecule.
"To capture as much carbon as possible, you want the longest chain hydrocarbons. Chains with 8 to 12 carbon atoms would be the ideal," says Cargnello.
Current technologies struggle to get near this goal. The longer the chain, the more heat and pressure are required, making the process less efficient and more expensive.
Cargnello and his team focused their research on organic polymers – materials with pores that can be scaled easily to suit the kind of structure needed to maximize the reactivity of carbon dioxide and hydrogen into chains.
Combined with an effective catalytic metal to hasten the process (in this case, the element ruthenium combined with titanium oxide), the researchers could improve efficiency by modeling the polymer's pore structures.
And improve it they did, demonstrating a 10 fold increase in the turnover of high molecular weight carbon chains.
Remarkably, the rate of production for the four-carbon-long chains of butane ramped up 1,000 fold by coating the catalyst in a specific kind of porous organic polymer.
"An uncoated catalyst gets covered in too much hydrogen on its surface, limiting the ability of carbon to find other carbons to bond with," says chemical engineer Chengshuang Zhou, a doctoral candidate in Cargnello's lab.
"The porous polymer controls the carbon-to-hydrogen ratio and allows us to create longer carbon chains from the same reactions."
Ideally, any carbon we could pull from the atmosphere should be locked away for good. Carbon neutral fuels might help address the economics of making such technology pay for itself, at the expense of simply creating a turn-style for atmospheric carbon.
The step to churning out more butane, which is less likely to vaporize rapidly into a gas at room temperature and leak into the atmosphere like smaller chains, is a significant one.
Still, with further research, this kind of chemical process just might produce more stable forms of hydrocarbon that could be incorporated into more persistent structures we'd want to store away safely.
Of course nature's solution to plucking out carbon and packing it into biological materials is always going to be a number one priority. Nothing beats locking up CO2 in the form of plants.
But if this helps us wean ourselves off our dependency on fossil fuels sooner rather than later, it's a solution well worth pursuing.
This research was published in Proceedings of the National Academy of Sciences.