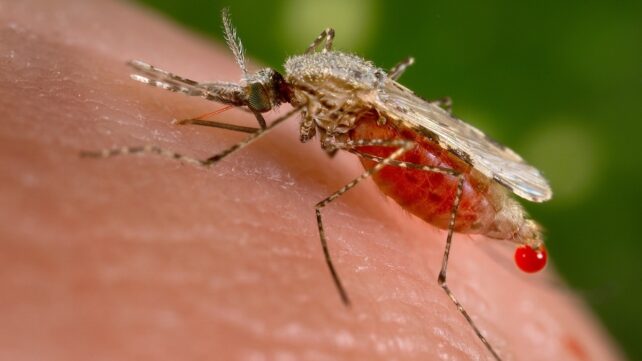
Malaria killed about 610,000 people globally in 2024, with most deaths occurring in Africa, where young children remain the most vulnerable. Now, scientists report a potentially game-changing weakness they've found in the parasites that cause the disease.
The findings provide insights into these parasites' complex biology, the authors of the new study explain, and may help identify new ways to sabotage them.
Early forms of malaria-like parasites date back to the Cretaceous, and despite the advent of vaccines, the ancient scourge still plagues humans across ever larger regions of the Earth.
Along with ongoing vaccine development, researchers are searching for any vulnerabilities to exploit in the parasites or the mosquitoes that transmit them to humans.
"What makes this discovery so exciting is that the malaria parasite's 'Aurora' complex is very different from the version found in human cells," says senior author Rita Tewari, a parasite cell biologist at the University of Nottingham.
Malaria is caused by protists, single-celled eukaryotic organisms that are not classified as animals, fungi, or plants.
These specific protists belong to the genus Plasmodium, which includes more than 150 named species that infect a range of vertebrates. Only five species cause malaria in humans, but their impact is devastating.
Plasmodium parasites replicate quickly within humans and in Anopheles mosquitoes. Clarifying the details of this swift process could be invaluable to our efforts to disrupt it.
Mitosis in malaria parasites is "fundamentally different," the study's authors write. The single-celled creatures divide and grow in a unique way, unlike the typical process seen in human cells and in many other eukaryotic organisms.
The new study focuses on a protein called Aurora-related kinase 1 (ARK1), a vital component in Plasmodium parasites' unconventional cell division and growth.

ARK1 helps direct traffic during the parasites' mitosis, the researchers explain, and bears responsibility for organizing a specialized apparatus called a spindle. This molecular equipment is critical for properly separating genetic material to produce new parasites.
Using genetic engineering techniques known as conditional gene knockout and gene knockdown, the researchers inactivated ARK1 in Plasmodium parasites to investigate its role.
The results suggest ARK1 is like an Achilles' heel for malaria parasites. Without this protein, the parasites failed to form effective spindles, leading to unsuccessful replication.
Parasites lacking ARK1 failed to complete their development in vertebrate host cells or mosquitoes, preventing them from spreading disease.
Given the importance of ARK1 for malaria parasites, this protein makes an enticing target for new antimalarial interventions, the researchers note.
"The name 'Aurora' refers to the Roman goddess of dawn, and we believe this protein truly heralds a new beginning in our understanding of malaria cell biology," says co-first author Ryuji Yanase, a cell biologist at the University of Nottingham.
And thanks to fundamental differences in our cellular machinery and mechanics, targeting ARK1 might offer a way to neutralize malaria parasites with minimal harm to their human hosts.
"This divergence is a huge advantage," Tewari explains.
"It means we can potentially design drugs that target the parasite's ARK1 specifically, turning the lights out on malaria without harming the patient."
Related: Compound That Turns People Yellow Could Protect Against Malaria
By detailing the unorthodox replication techniques these parasites employ, including the pivotal role of ARK1, the authors hope to provide a foundation for future research that explores new ways to thwart this ancient nemesis.
The study was published in Nature Communications.