Sleeping with a clunky breathing machine is a problem for many with sleep apnea, but a more comfortable alternative is on the horizon.
In a recent clinical drug trial that ended in 2023, participants who took the highest daily dose of an epilepsy drug had nearly 50 percent fewer breathing interruptions while sleeping.
"We have been working on this treatment strategy for a long time, and the results show that sleep apnea can indeed be influenced pharmacologically," says Jan Hedner, who specializes in pulmonary medicine at the University of Gothenburg in Sweden.
"It feels like a breakthrough, and we now look forward to larger and longer studies to determine whether the effect is sustained over time and whether the treatment is safe for broader patient groups."
Sleep apnea can occur when the upper airways suddenly collapse during periods of rest, leading to obstructed breathing patterns that deprive the body of oxygen and regularly disrupt sleep.
CPAP (continuous positive airway pressure) machines are designed to keep the upper airway open throughout the night. For many years, this has been the standard and most effective way to improve sleep in patients.
At the very end of 2024, however, the US Food and Drug Administration (FDA) approved a drug called tirzepatide (brand names Mounjaro or Zepbound) for adults with obesity and moderate-to-severe sleep apnea.
This was the first medicine to be approved for the airway condition, but its sleep-related benefits primarily seem to stem from weight loss in the neck, creating a larger airway that is less prone to obstruction.
This medication does not treat other underlying issues of obstructive sleep apnea, such as muscle strength in the airways or the body's respiratory control.
Hedner and colleagues argue "there is a clear, unmet need for an effective and well-tolerated treatment" that is more direct.
The anticonvulsant drug they are testing is called sultiame. It was first synthesized in the 1950s, and while it is now sold in several European nations, as well as Israel, Japan, and Australia, to treat partial seizures, it has never been registered in the US.
In a preliminary study in 2024, researchers in Europe found that sultiame could improve upper airway muscle tone and stabilize respiratory control.
Building on these results, researchers at the University of Gothenburg have now conducted a large, stage II clinical trial to find an appropriate dosage for sleep apnea.
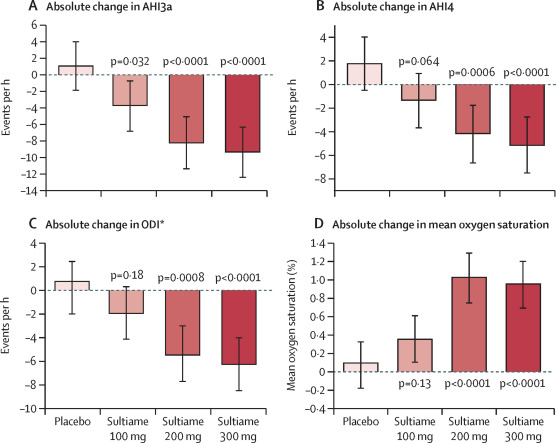
They split a cohort of 240 participants into four groups: one took a placebo daily, and the other three groups took either 100 mg, 200 mg, or 300 mg of sultiame daily.
Participants included in the trial hailed from medical institutions in five European countries (Belgium, Czechia, France, Germany, and Spain), and they had all been diagnosed with moderate to severe obstructive sleep apnea.
Those who took any dose of sultiame an hour before bed showed fewer breathing disturbances, better overnight oxygenation, and reduced excessive daytime sleepiness, compared to the placebo group.
Those taking the two highest doses of sulthiame, however, reaped the most benefits. Doses of 200 mg or 300 mg reduced sleep apnea severity by approximately 30 to 50 percent, the trial researchers report, far outperforming the placebo.
There were no clinically relevant safety concerns, but because side effects increase with the dosage, 200 mg seems to be the sweet spot.
"These findings introduce a new individualized pharmacotherapy in patients with sleep disordered breathing," conclude Hedner and colleagues.
Sleep apnea is estimated to affect millions of people worldwide and is linked to a host of negative health effects, including cognitive issues, cardiovascular disease, and insulin problems. Some studies suggest that the risk of developing the condition is set to increase by nearly 50 percent in the coming years.
Effective treatments are few and far between. Taping the mouth shut may worsen sleep apnea, researchers say, and even some mouth guards that push the tongue forward have downsides to consider, such as changing a person's bite.
Related: Taping Your Mouth Shut For Sleep Apnea Is Not a Good Idea
Other researchers have tried using electrical pulses on the tongue to keep the upper airways open, and some recent studies have even found that blowing a conch shell can help, too.
A drug that can address the root causes of sleep apnea would be a welcome addition to the mix, with the potential to make treatments cheaper, more accessible, and more comfortable the world over.
The study was published in The Lancet.

