Unfortunately, for those of us approaching or beyond 30 years of age - this is the point our muscles start to basically shrink away. You may think you've heard it all before, but a new review presents yet another reason we really should avoid slacking off when it comes to moving our behinds.
Amongst many other things, exercise has been shown to increase mitochondria - energy-generating organelles - within cells; it also increases blood supply, waste removal, and bone strength.
While many muscle degeneration studies focus on much older people, the slow decline of muscle tissue in our bodies actually begins in our 30s, with an average of between three to five percent muscle loss every year.
Thankfully, our muscles are able to regenerate, so with a bit of effort on our part, we can counteract this wastage.
Muscle growth, or regeneration after injury, involves satellite cells. When they divide into a new generation of cells, they can either become more satellite cells, or myoblasts - cells which then divide into muscle cells. When they're not used however, satellite cells go into an energy-saving dormancy.
Cellular biologist William Chen and colleagues from the University of Ottawa in Canada reviewed the literature on how these cells function, including human strength training and endurance exercise studies, as well as molecular and genetic studies on mice.
"It is evident that satellite cells are activated through various signalling pathways following exercise," the team found, with multiple studies of human training sessions indicating an increase in satellite cell proliferation.
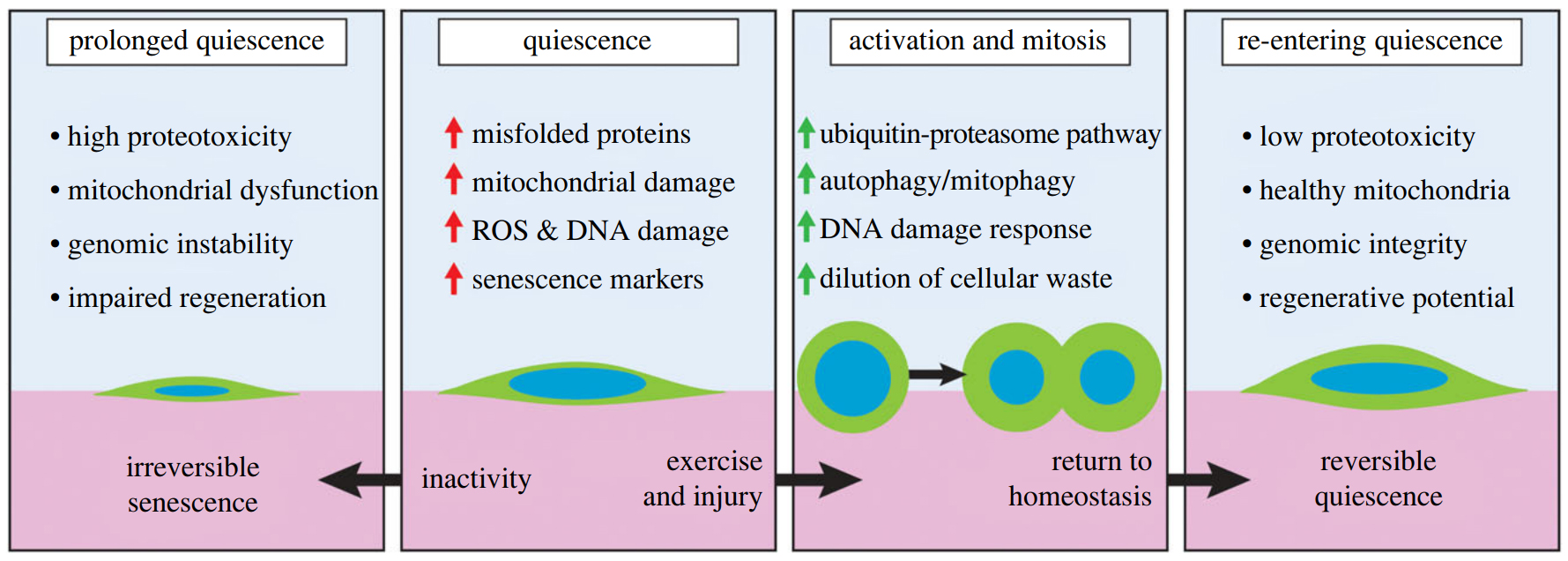
Neglecting exercise causes problems, because like all cells, satellite cells become damaged and filled with cellular trash as the normal processes of life batter them about as they age. Even while dormant, when cellular activity is reduced, the process of keeping themselves in this state still produces cellular waste.
Thus, if satellite cells stay dormant for too long, the waste build-up can cause damage, preventing the cells from dividing and replacing themselves. Even worse, in ageing muscles, if they're not activated, satellite cells can get stuck in the dormant phase and lose all ability to divide and multiply.
This means they are no longer capable of replacing defunct muscle cells with shiny new healthy ones, or making more copies of themselves.
 Healthy dormancy (quiescence) requires regular activation. (Chen et al, Open Biology, 2020)
Healthy dormancy (quiescence) requires regular activation. (Chen et al, Open Biology, 2020)
"Physical inactivity becomes more detrimental with age as satellite cells accumulate higher levels of cellular waste and DNA damage during longer periods of deep quiescence," Chen and colleagues explain in their review.
So, giving these cells a regular prod through exercise, to remind them how to be active, helps more of them retain their full function.
However, "exercise alone does not prevent all age-related defects in skeletal muscle," the team wrote.
"The satellite cell population still diminishes to some degree over age… Regardless, lifelong resistance exercise should be pursued as the effects of [age-related skeletal muscle loss] are greatly diminished when compared with sedentary lifestyles."
The researchers point out the exact mechanisms of satellite cell replacement are not yet fully understood, nor is their behaviour during exercise. They recommend more research be focused in these directions.
With skeletal muscle making up 40 percent of our body in terms of weight, it makes sense that the condition of the bulk of these cells and their makers has huge impacts on our overall health.
Muscle strength is correlated with increased quality of life, particularly as we age. So this aspect of satellite cell biology is just one of many ways exercise helps to keep our muscles and the rest of us healthy.
This review was published in Open Biology.