Brain aneurysms are the stuff of nightmares – a blood vessel in your brain which silently bulges or balloons, with the risk of it one day rupturing, causing life-threatening complications.
Although there are a number of treatments for brain aneurysms, you can imagine that blood vessels in the brain can be a little tricky to reach. So, for the purposes of evaluating treatments and giving doctors some hands-on training time, some kind of substitute before they get inside your brain would be ideal.
A team of researchers based in the US has now brought such a thing into reality. They produced the first living, bio-printed aneurysm outside the human body, conducted a medical procedure on it, and then observed it healing.
If doctors notice that someone has a brain aneurysm before it ruptures – unfortunately they're hard to spot so this doesn't always happen – they'll attempt to stop blood flow to the area. There are a couple of different ways to treat it, and none of the options are particularly fun.
The first option is surgical clipping – where a surgeon removes part of your skull and puts a tiny metal clip on the base of the aneurysm.
The other option is something called endovascular coiling, where a catheter is inserted into an artery in the groin, and then threaded through the body to the aneurysm. A coil is then pushed through the catheter and into the aneurysm.
Both treatments stop or disrupt blood flow, which means the aneurysm won't get any larger and potentially burst. But both methods also present their own issues, and it can be hard to pick which option is the best for any given patient.
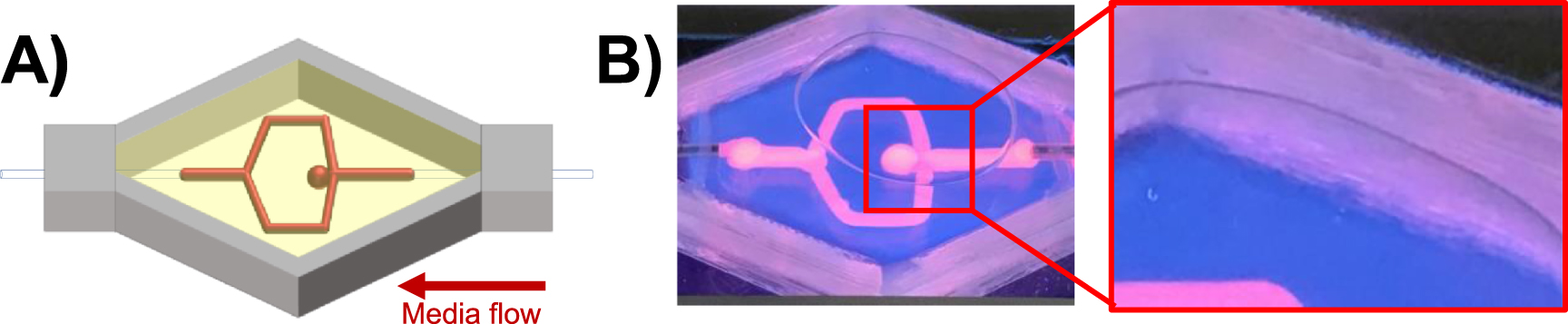 (a) Illustration of the 3D-printed aneurysm. (b) The aneurysm vessel structure. (Kang et al., Biofabrication, 2020)
(a) Illustration of the 3D-printed aneurysm. (b) The aneurysm vessel structure. (Kang et al., Biofabrication, 2020)
"We looked at the problem and thought that if we could pair computational modelling and experimental approaches, maybe we could come up with a more deterministic method of treating aneurysms or selecting treatments that could best serve the patient," said Lawrence Livermore National Laboratory engineer and senior author William Hynes.
"Now we can start to build the framework of a personalised model that a surgical practitioner could use to determine the best method for treating an aneurysm."
The team 3D-printed an aneurysm-shaped structure using gelatin-fibrin hydrogel, and then carefully seeded the scaffold with human brain cells called hCMECs (human cerebral microvascular endothelial cells). These cells spread out, lining the aneurysm over the next seven days, forming a living 3D-printed aneurysm.
Although fake aneurysms like this have been created before, this is the first time human cells have been used to create a living structure.
Once the cells had spread, the team then went to work experimenting on their newly formed aneurysm by flowing cow blood plasma through the structure and then doing their own endovascular coiling. Excitingly, the coil successfully formed a clot at the site – meaning blood flow was disrupted, and the model worked!
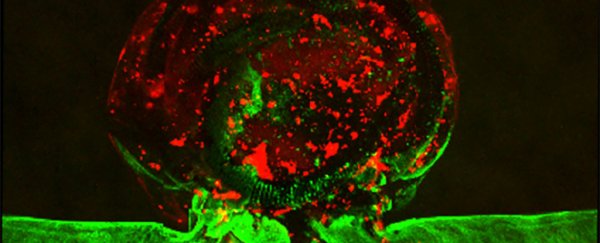
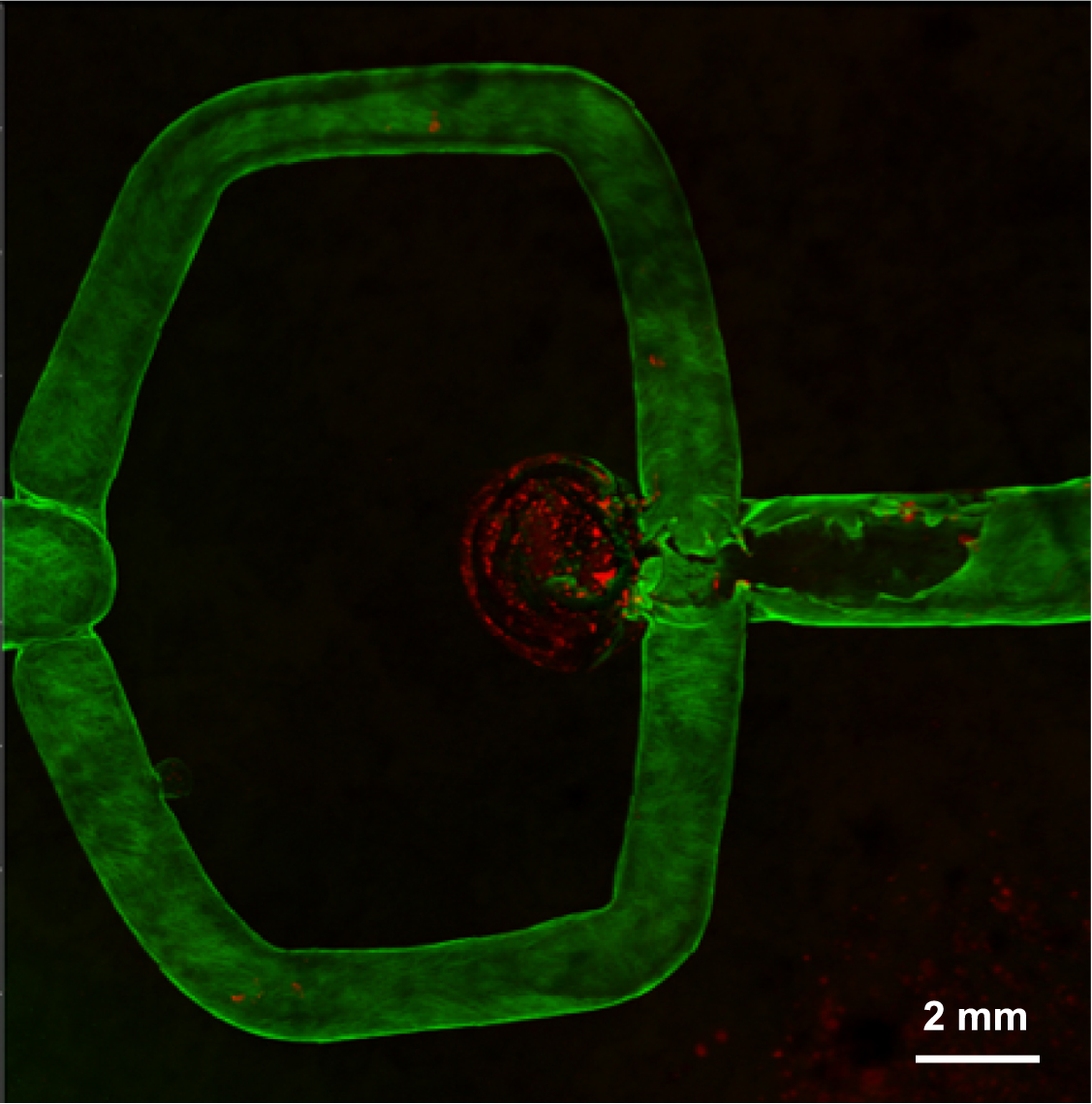 Clot, in red, and endothelial cells, green, in the aneurysm model. (Kang et al., Biofabrication, 2020)
Clot, in red, and endothelial cells, green, in the aneurysm model. (Kang et al., Biofabrication, 2020)
The team stresses that there's still a long way to go before this is ready for doctors. They still have to work more on computer models of clotting in three dimensions, using better particle image velocimetry, and becoming better at mimicking the wall shear stress that causes aneurysms in the first place.
But with the help of patient brain scans and computer modelling systems, they hope the physical models can go some ways towards being personalised to a specific patient's aneurysm.
This is an exciting development that has a lot of potential uses, and the team thinks that this could be a better option for evaluating new treatments than animal models, and when used with computer modelling, it could cut the time it takes for new surgical techniques to get to the clinic.
"This in vitro model can be used for training surgeons or medical students to deploy neurovascular devices into a complex vascular structure," the team writes in their paper.
"[It] can be readily utilised in investigations related to analysing clotting responses of embolisation devices, healing responses after treatment, and the biophysical mechanisms of aneurysm formation or rupture using the resulting haemodynamic data."
The research has been published in Biofabrication.