Researchers from Japan have worked out how to make human embryonic stem cells organise themselves into functioning, cerebellum-like brain tissue. This is an important step towards growing healthy brain tissue that could help heal patients with brain damage or neurodegenerative diseases.
So far, while scientists have managed to grow stem cells into other organs, such as the stomach and even the penis, this process has been particularly tricky for the nervous system, because researchers not only need to generate particular cell types, they also have to make these cells types connect to each other in very specific ways.
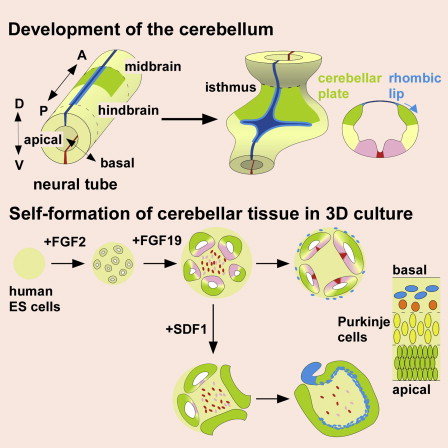
Now, a team of researchers from the RIKEN Centre for Developmental Biology in Japan have overcome this challenge. By carefully applying signalling molecules to 3-D cultures of human embryonic stem cells, they've managed to not only trigger them to differentiate into functioning cerebellum cell types, but also got them to organise themselves into the multi-layer, accordian-like structure found in the naturally developing cerebellum.
The cerebellum is the region at the bottom of the brain that plays a role in motor control, and is involved in cognitive functions including attention and language. The most important cell types of the cerebellum are Purkinje cells and granule cells, and the team managed to develop both of these using embryonic human stem cells.
The work builds on the team's previous research in mice, which allowed them to map the hormones and growth factors that are required during embryonic growth in order for the cerebellum to develop properly. They've now used this information to create a "recipe" that they applied to human embryonic stem cells in order to trigger the development of cerebellum tissue in the lab.
"The principles of self-organisation that we have demonstrated here are important for the future of developmental biology," said study leader Keiko Muguruma in a press release.
Publishing in Cell Reports, the team describes how they first treated human embryonic stem cells with fibroblast growth factor 2, which, after three weeks, caused the stem cells to differentiate into neurons from the midbrain/hindbrain region, where the cerebellum is located.
After five weeks, the stem cells were displaying signs of differentiating into the cerebellar plate neuroepithelium - the part of the nervous system that turns into the cerebellum.
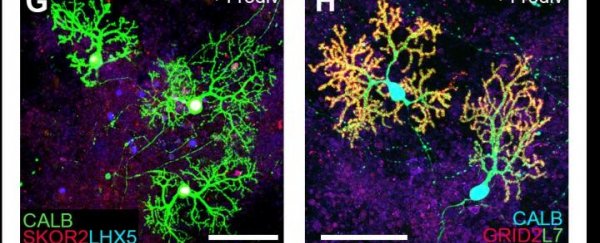
At around 15 weeks, they found that cells were expressing markers for late Purkinje cells (shown above), which suggests that they'd differentiated into this important and specialised neuron type. Electrophysiological recordings showed that these cells even responded to electrical currents in the same way that they would expect functioning cerebellum cells to respond.
They then built on this research and added a range of different growth factors at different stages during the differentiation process in order to get these cerebellum cells to organise themselves into the layered structure of the cerebellum tissue in the human brain.
Knowing this recipe will help researchers who are hoping to be able to create healthy cerebellum tissue out of their patients' reprogrammed adult stem cells - a development that could help treat diseases such as spinocerebellar atrophy, which is a degenerative genetic disease that affects people's ability to speak, walk and move properly.
You can see a comparison of how the cerebellum normally develops in the human brain, compared to how it developed in the lab, in the illustration below. It's complicated science, but it's really exciting to see researchers finally begin to unlock and replicate brain development, as it's the first step to being able to treat a whole range of new diseases.