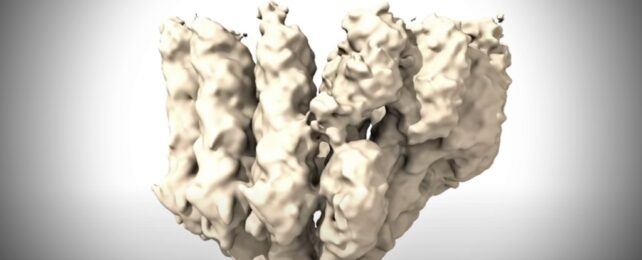
A team of researchers in Spain has achieved a breakthrough by capturing the world's first detailed images of a human cell's 'highway network' beginning to emerge.
The high-resolution images and atomic-scale film help explain a longstanding puzzle of how small structures called microtubules form during cell division. The discovery could progress the development of targeted treatments for cancer, and many other conditions.
"Microtubules are critical components of cells, but all the images we see in textbooks describing the first moments of their creation are models or cartoons based on structures in yeast," says biochemist Thomas Surrey from the Centre for Genomic Regulation in Barcelona.
"Here we capture the process in action inside human cells. Now that we know what it looks like, we can explore how it's regulated. Given the fundamental role of microtubules in cell biology, this could eventually lead to new therapeutic approaches for a wide range of disorders."
In a cell, microtubules act like highways for transporting components between different areas. They also play a crucial role in cell division, acting like lassos by hooking and separating duplicated chromosomes, allowing the cell to divide into two.
Through a process called microtubule nucleation, microtubules are built out of proteins called tubulins by a structure called a gamma-tubulin ring complex (γ-TuRC), which acts like a blueprint guiding their formation.
A mystery arose when it was found that human γ-TuRC exposes fourteen rows of tubulins instead of the expected thirteen that microtubules need for cells to function correctly, and γ-TuRC's ring structure appeared open when scientists expected it to be closed.
The first high-resolution visuals of both γ-TuRC and microtubules together help to unravel this mystery. The team had to slow down the high-speed process to see it in the lab.
"We had to find conditions that allowed us to image over a million microtubules in the process of nucleation before they grow too long and obscure the action of γ-TuRC," says cell biologist Cláudia Brito from the Centre for Genomic Regulation.
"We were able to achieve this using the molecular toolbox of our lab and then freeze the microtubule stubs in place."
The researchers used cryo-electron microscopy and some complex image processing to determine the 3D structure of γ-TuRC during microtubule formation. They found that as γ-TuRC initiates nucleation and the microtubule grows, it undergoes a shape change.
It cleverly closes, stowing away one tubulin so it matches the microtubule's design. A new latch mechanism was discovered too, and it revealed that the growing microtubule assists the γ-TuRC template in finding its correct shape.

"We have visualized the process that initiates microtubule formation, and we see that human γ-TuRC is an open ring that closes to effectively become a perfect template to nucleate microtubules," says structural biologist Oscar Llorca from the Spanish National Cancer Research Centre.
"But we also discovered that this ring, in order to close, needs the 'first brick' of a microtubule to be put in place; when this happens, a region of the human γ-TuRC acts as an anchor that engages this 'first brick' to then close the ring and launch the formation of the microtubules."
Problems with microtubules can cause many health complications, including cancer. Microtubule dysfunction can also exacerbate conditions like asthma and heart disease, as well as contribute to neurodegenerative disorders.
Although microtubules are a target for some cancer treatments, these drugs have the unfortunate side effect of affecting healthy cells too. And tumors have the potential to become resistant to these medications with time. So finding out more about microtubules and their construction process is valuable.
"As further work clarifies how regulators bind to γ-TuRC and how they affect the conformational changes during nucleation, it may transform our understanding of how microtubules work," Surrey explains, "and eventually offer alternative sites that one might want to target to prevent cancer cells from going through the cell cycle."
The study has been published in Science.