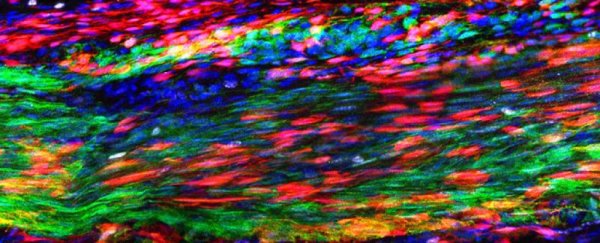
Tendon injuries are notoriously painful and slow to heal, and for a long time scientists haven't fully understood why this connective tissue struggles to regenerate itself when it gets hurt.
Fortunately, a new discovery made by researchers in the US could lead to entirely new ways to promote healing in this injury-prone part of our bodies, thanks to the identification of a previously unknown kind of tendon stem cell.
"Because tendon injuries rarely heal completely, it was thought that tendon stem cells might not exist," says developmental cell biologist Tyler Harvey from the Carnegie Institution for Science.
"Many searched for them to no avail, but our work defined them for the first time."
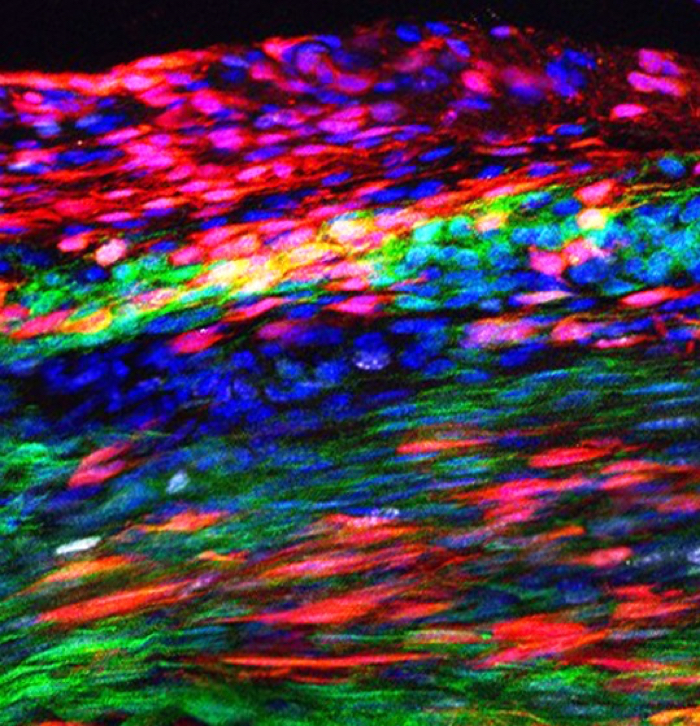 Red marks are newly discovered tendon stem cells that have self-renewed in the Patellar tendon after injury. (Tyler Harvey)
Red marks are newly discovered tendon stem cells that have self-renewed in the Patellar tendon after injury. (Tyler Harvey)
It's not the only instance of scientists investigating the existence of tendon stem cells, but it's the only one to identify a newfound group of cells called Tppp3+ (short for tubulin polymerisation-promoting protein family member 3-expressing cells) in the Patellar tendon, below the kneecap.
According to the researchers, who made the discovery in mice, these progenitor cells can generate new tendon cells called tenocytes and self-renew upon injury.
That's important, because when tendons become injured, their fibrous function – to connect muscle to bone – becomes impaired, due to tears in the tissue that cause pain, stiffness, swelling, and inflammation – not to mention a stubborn resistance to getting better.
"Tendon injuries cause prolonged disability and never recover completely," Harvey and his co-authors explain in their new paper.
"Injuries to tendons represent the worst clinical outcome within the musculoskeletal system, as healed tendons rarely attain the integrity of the undamaged state."
That's why the Tppp3+ discovery could be a big deal, as it's possible we could use these stem cells to enhance natural tendon regeneration, tardy as it is.
What can hold that healing process back, the results show, is when a receptor called platelet-derived growth factor receptor alpha (Pdfgra, which is expressed by some Tppp3+ cells), becomes inactivated.
The receptor is usually activated by a protein called PDGF-AA, generating tenocytes which repair the tendon. Scar-forming fibrotic cells are also activated by this same protein.
When the receptor can't be activated on TPPP3+ cells, tendon regeneration halts, while scar-formation continues uninterrupted.
"Tendon stem cells exist, but they must outcompete the scar tissue precursors in order to prevent the formation of difficult, fibrous scars," says one of the Carnegie team, Chen-Ming Fan.
"Finding a therapeutic way to block the scar-forming cells and enhance the tendon stem cells could be a game-changer when it comes to treating tendon injuries."
Of course, to do that, we first need to see these Tppp3+ results replicated in humans, and then figure out a way to inactivate Pdfgra safely, without affecting other cell functions.
Whether all that can be achieved remains to be seen, but if it can, we could be on the verge of new therapeutic options for tendon injuries – something of an Achilles heel itself in terms of medical treatments available.
"Even though the complex contribution of the microenvironment requires further elucidation, the latest work … represents an important step forward in the study of tendon regeneration and the cellular and molecular mechanisms involved," reconstructive surgeons Ashley Titan and Michael Longaker, both of Stanford University, write in a commentary on the research.
The findings are reported in Nature Cell Biology.