Scientists have created a first-of-its-kind mouse model of the COVID-19 disease, capable of revealing how the SARS-CoV-2 virus wreaks havoc on multiple organ systems in the animal's body.
While this experimental model doesn't directly correspond to human coronavirus cases, it's a breakthrough in other ways, giving us a potential test system for exploring how the deadly respiratory pathogen extends way beyond the respiratory system in many cases of infection.
"This mouse model is a really powerful tool for studying SARS-CoV-2 in a living system," explains cardiologist Arjun Deb from UCLA.
Mouse models of COVID-19 have been engineered before, but none have gotten us to this point, the researchers say, by demonstrating what extra-pulmonary manifestations of COVID-19 look like.
 (JCI Insight/UCLA Broad Stem Cell Research Centre)
(JCI Insight/UCLA Broad Stem Cell Research Centre)
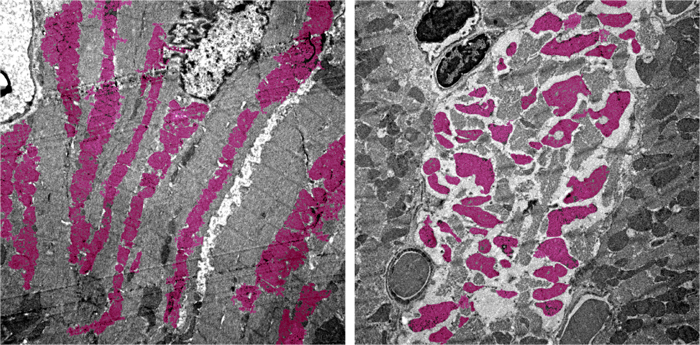
Above: Heart muscle cells in an uninfected mouse (left) and a mouse infected with SARS-CoV-2 (right), with mitochondria seen in pink.
That's been a significant limitation for animal-based research into the virus's progression, and it's due to a couple of reasons. While mouse cells contain an analogue of the ACE2 receptor that the SARS-CoV-2 uses to bind to human cells, the virus doesn't seem to attach to the mouse version of the molecule.
Genetically engineering mice with the human version of ACE2 provides a workaround of sorts, but before now, scientists hadn't succeeded in inducing multiple organ failure in mice, which could mimic the way human cases of extra-pulmonary infection tend to present.
The shortfall may have been because prior studies used nasal inoculation on mice, infecting the animals with SARS-CoV-2 through the nose, which doesn't seem to develop into full-blown, system-wide coronavirus infections in mice.
In the new study, led by first author Shen Li, a cardiologist at UCLA, the team got around this by injecting the virus into the bloodstreams of engineered mice, where it could reach the human version of ACE2 (called hACE2) in the heart and other vital organs.
Unfortunately for the subjects, the experimental tweaks worked. And while it wasn't a good outcome for the mice involved, being able to study systemically induced toxicity in SARS-CoV-2-infected mice could significantly expand our knowledge of the virus's branching capabilities in human cases. Such knowledge is sorely needed.
"Among COVID-19 patients, those who have organs involved other than the lungs are most at risk of a bad outcome," Deb says.
"So we felt it was really important to understand how the virus affects those other organs."
In the experiment, systemic administration of the infection provoked rapid results.
Within seven days, the infected mice "demonstrated profound morbidity, severely restricted activity, and were found huddled at the corner of the cage", in contrast with a control group of similarly engineered mice who were spared the infection, remaining healthy.
In the same amount of time, the infected mice lost up to about 25 percent of their body weight due to sharply reduced food consumption, necessitating euthanisation.
The infected mice also had damaged spleens, irregular heart activity and blood pressure, and altered levels of immune cells – all symptoms resembling human cases of COVID-19.
After the animals were euthanised, analysis of their organs revealed changes in gene expression in multiple tissues, disrupting cellular processes that generate energy in the body.
"If a virus snuffs out the energy-generating pathways in multiple organs of the body, that's going to really wreak havoc," Deb explains.
Beyond these effects, the infected mice also bore numerous signs of epigenetic changes, which could explain the altered gene expression evident in multiple organs.
It's not known for sure, but the impacts of this could potentially be felt long after an infection has been beaten by the immune system – and, hypothetically speaking, could be the basis of the prolonged symptoms experienced by COVID-19 'long haulers'.
"Although the physiological significance of SARS-CoV-2 altered DNA methylation patterns is not clear from our study, our model provides proof of concept that such epigenetic changes do occur soon after SARS-CoV-2 infection and can potentially led to persistent transcriptional changes affecting tissue homeostasis and organ function," the authors write in their paper.
"Such epigenetic changes potentially occurring in humans with COVID-19 could lead to symptoms from persistent changes in dysregulated gene expression in infected tissues even in the absence of tissue viral burden."
Again, that's all depending on how much crossover really exists between artificially induced mouse COVID-19 and all-too-easily caught human COVID-19.
That relationship is yet another unknown, although thanks to this grisly model, at least now we can start asking the questions.
The findings are reported in JCI Insight.