Graphene is a common, two-dimensional material made of carbon. When stacked in layers it's known as graphite, a dark substance most people come across mixed with clay in so-called 'lead' pencils.
Thanks to its unique mix of structural, thermal, and electromagnetic properties, graphene has attracted a great deal of attention for its potential role in a diverse range of future technologies.
Every atom of carbon in a graphene sheet is tightly bound to three other atoms at identical angles, forming a flat, honeycomb-like structure. Similar to diamond – which is a three-dimensional carbon crystal where every atom is connected to four neighbours – these strong bonds lend the structure significant robustness.
Specifically, graphene has incredible tensile strength, especially on a small scale. This means that compared to a thin string of crystalised steel just a few micrometres across, graphene is more than six times harder to rip apart. Tests with other, less ideal forms of steel have previously suggested it could be hundreds of times stronger.
The basic hexagonal shape of graphene forms the basis of a fullerenes, which is a hollow molecular structure made up of 60 or more carbon molecules, like those beautiful carbon spheres called 'buckyballs'. The nanoscale mesh on these structures is so flexible, it can be rolled into hollow cylinders, making them potentially useful containers on a molecular scale.
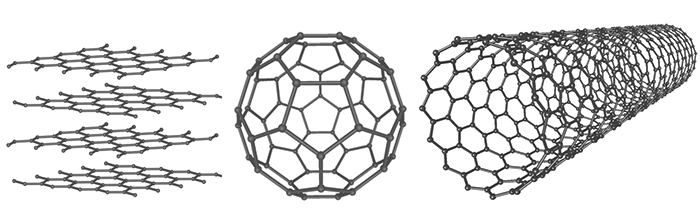 Graphene layers, a bucky ball, and a nanotube (mstroeck/Wikimedia commons/GFDL)
Graphene layers, a bucky ball, and a nanotube (mstroeck/Wikimedia commons/GFDL)
Since the element carbon has four available electrons for bonding, each atom in a sheet of graphene contributes at least one unbound electron to the molecule. That idle negative charge helps give the material incredible conductive and superconductive properties, which makes it suitable for advanced electronics.
Add to these talents its impermeability to many liquids and gases and an ability to repel water, as well as an incredible transparency, and it's easy to see why graphene has been described as a 'wonder material' to watch out for.
How was graphene discovered?
While the basic structural properties of graphite were determined experimentally in the early 20th century, the first real theory on graphene's unique chemistry wasn't worked out until 1947, when Canadian theoretical physicist Philip Russell Wallace tried to come up with ways to explain graphite's electrical properties.
As interesting as the material seemed to be, forcing graphite to shed more than a few single sheets at a time was challenging.
In 2004, University of Manchester researchers Andre Geim and Kostya Novoselov famously devised a way to produce sufficient quantities to study, by peeling graphene from graphite using little more than sticky films of scotch tape. They've since earned a Nobel Prize for their groundbreaking studies on this material.
Future use of graphene will rely on far more efficient production methods, inspiring researchers to look for innovative new ways to make vast sheets of flat, 2D carbon mesh.
All topic-based articles are determined by fact checkers to be correct and relevant at the time of publishing. Text and images may be altered, removed, or added to as an editorial decision to keep information current.
