There are all kinds of pollutants in the air around us. Outdoors, these can be washed away through the falling rain, and the oxidation that happens after ultraviolet light from the Sun interacts with ozone and water vapor.
So, what happens indoors?
As a new study shows, there's also some oxidation going on indoors too: the chemical cleaning that occurs via these hydroxyl (OH) radicals – short-lived reactive species whose job is to oxidize other molecules – happens through a combination of ozone leaking in from the outside, and from the oxidation fields that we create around ourselves.
In some scenarios, levels of OH radicals indoors are comparable to daytime outdoor levels, scientists have found. In other words, we're walking, breathing, chemical reaction machines, which has implications for indoor air quality and human health.
"The discovery that we humans are not only a source of reactive chemicals, but we are also able to transform these chemicals ourselves was very surprising to us," says atmospheric chemist Nora Zannoni from the Institute of Atmospheric Sciences and Climate in Italy.
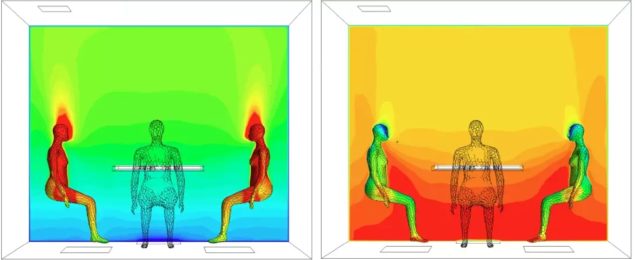
The team carried out experiments with three separate groups of four people in a special climate-controlled chamber, with levels of ozone that matched the upper end of what you might typically find indoors. Records were made of OH values both with and without ozone present, and before and after humans entered the room.
Through a combination of computational fluid dynamics modeling and actual air measurements (in part involving mass spectrometry techniques), it became clear that OH radicals were present, abundant – and forming around the human beings.
The scientists found that our personal oxidation fields are generated as ozone reacts to the oils and fats on our skin – particularly on the unsaturated triterpene squalene compound that makes up about 10 percent of the lipids that protect the skin and keep it supple.
"The strength and shape of the oxidation field is determined by how much ozone is present, where it infiltrates, and how the ventilation of the indoor space is configured," says Zannoni.
It's thought that we spend around 90 percent of our time indoors, and these findings have important implications for making sure that time is spent breathing air that's as clean and as healthy for us as possible – something we're all now acutely aware of, thanks to the pandemic.
While we always knew that oxidation processes were happening indoors, it seems that in some conditions, the reactions generated by humans are the dominant ones.
It's important to understand these processes, both in isolation and in relation to other indoor chemicals that might arise from building materials, furnishings, and scented products, because reactions could produce respiratory irritants as well as removing pollutants.
There's still a lot more work to do, as well: the scientists are keen to understand how humidity levels affect the reactions, for example, and how more and more people inside a room might change the picture.
Further, there is a possibility that the oxidation fields humans produce might even affect our perception of odor.
"We need to rethink indoor chemistry in occupied spaces because the oxidation field we create will transform many of the chemicals in our immediate vicinity," says atmospheric chemist Jonathan Williams, from the Max Planck Institute for Chemistry in Germany.
The research has been published in Science.