Physicists in the US say they have created a simulation that, for the first time, demonstrates "ideal glass" is possible, resolving a decades-old paradox.
In some ways, the kind of glass you drink iced water from has more in common with the liquid than the frozen crystals floating within it. Its molecules are jumbled like those in a liquid, a random structure from just one of countless possible arrangements.
An ideal glass is different. What would appear to be a jumbled mess of molecules would have no other options - or, as it's known in physics, a minimal entropy.
This type of glass was first discussed in 1948 by chemist Walter Kauzmann, who proposed that because entropy drops as liquids cool and form glass, there could be a temperature low enough to eliminate it entirely. This would still be a random arrangement, but it would be packed so precisely that it could not be reconfigured in any other way.
In the decades since, the feasibility of such a paradoxically ordered glass has been extensively debated.
In the new study, University of Oregon physicist Viola Bolton-Lum and colleagues used computer models to show that ideal glass is possible in 2D – a glass with particles that have an amorphous arrangement but are also highly ordered and uniform, so it behaves like a perfect crystal.
"In addition to resolving a long-standing mystery, this methodology represents a valuable shortcut in the generation of well-equilibrated glassy systems," write the researchers in their published paper.
"The creation of such an ideal packing makes possible a complete exploration and explanation of two-dimensional jammed and glassy systems."
It was clear that normal cooling wouldn't work to reach the ideal glass state – that it would take an infinite amount of time. For their models, they introduced a cheat code of sorts that allows the glass particles to be resized as they're packed.
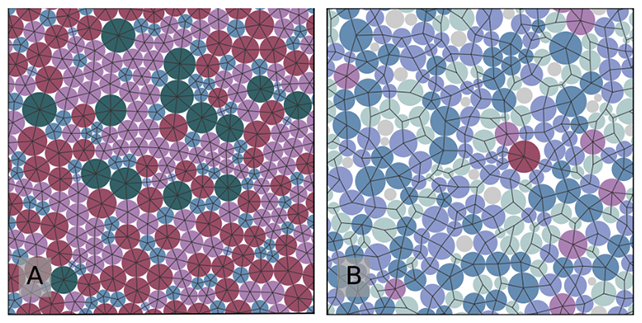
This extra degree of flexibility makes the difference, yielding a glass that appears amorphous yet exhibits crystalline properties. Specifically, the resulting glass is much more solid and stable than normal, with each particle having an average of six points of contact with a neighbor for that extra support.
"We think that we've hit upon a resolution, by showing that such a state is not a paradox at all," physicist Eric Corwin, from the University of Oregon, told Ingrid Fadelli at Phys.org. "Indeed, we can construct it."
One way the glass would differ from regular glass is in how it would react if hit. Rather than this causing the rather haphazard, messy vibrations of normal glass, ideal glass would vibrate with perfect uniformity – like a diamond, for example.
There would also be what's called hyperuniformity in the new material. When viewed up close, you wouldn't see any particle clumping or empty gaps, with each particle taking up just the right amount of space.
It's important to bear in mind that this research is theoretical: No one has yet manufactured ideal glass in the lab. In fact, the researchers acknowledge that standard heating and cooling processes won't be enough to create this glass – new approaches will need to be found before it can be produced.
However, the work does show that ideal glass isn't an impossibility, and – given its particular properties – it would likely be suitable for many varied purposes. What those purposes might be is hard to say, given it's still early days for imagining this material.

There is plenty for researchers to investigate regarding ideal glass and its development, particularly how the cheat code used in the simulations can be replicated in actual physical manufacturing processes. Given the advances in materials science, there is reason to believe ideal glass might one day exist.
Related: This Incredible New Bioplastic Could Be The Supermaterial of Tomorrow
"Novel approaches will be necessary to create such packings in practice, as they are not accessible through common thermal or mechanical processes," write the researchers.
"To create such systems in practice a physical implementation of our algorithm would have to be developed."
The research has been published in Physical Review Letters.

