Scientists have captured the fleeting moment when a heart starts beating, a feat made all the more remarkable when you consider the relative brevity of a zebrafish's life.
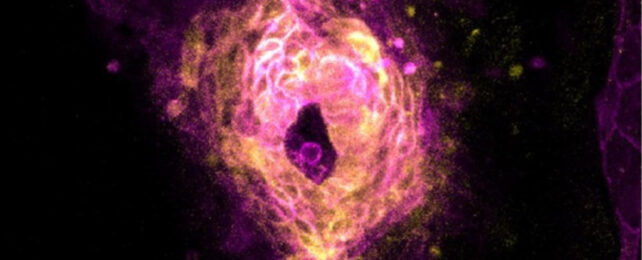
In a tight window about 20 hours into zebrafish development, the embryos' developing hearts jumped into action, emerging as one from an ensemble of single cells.
"It was like somebody had flipped on a switch," says Harvard University biophysicist Adam Cohen, senior author of the new study, which imaged zebrafish embryos snug in custom-made agarose molds to capture this once-in-a-lifetime event.

Using high-speed microscope imaging, the Harvard University researchers built on decades-old studies of first heartbeats in chicken, rats, and mice to peer into the hours-old zebrafish embryos and pinpoint the moment when heart cells begin to beat.
From past studies, it has become clear that the first heartbeat occurs even before the first heart structure, a primitive tube, forms. The first signs of activity are within populations of heart muscle cells, called cardiomyocytes, that scientists have found awash with calcium ions, but not in any organized way.
In the mature heart, calcium ions rush into and out of cardiomyocytes, generating action potentials (spikes of electrical activity) that trigger heart contractions.
Lead researcher Bill Jia along with Cohen and team continued that previous work by measuring how waves of calcium ions become organized and propagate in developing zebrafish embryos to trigger the heart's first, unified pulse.
"The heart beats about 3 billion times in a typical human lifetime, and it must never take a break," says Cohen. "We wanted to see how this incredible machine first turns on."
Slow, sparse calcium waves became larger and more frequent as developing cardiomyocytes assembled into a ring shape at the midline of the zebrafish embryos. Then, suddenly, calcium levels spiked and the heart cells released bursts of electrical activity that swept the tissue.
The first few heartbeats were a little irregular but soon settled into synchronized contractions. "A rhythmic, spatially structured beat arises well before connection to the circulatory system and blood pumping," the researchers observed.
Moreover, Jia and co. noticed how the zebrafish heart cells entered an excitable state about 90 minutes before the first heartbeat, as if readying themselves for action.
Interestingly, the surging waves of calcium ions preceding the first heartbeat didn't always originate in the same place in different zebrafish embryos, suggesting that there's nothing unique about the cells that fire first.
The so-called locus of initiation most often occurred in a central region of the cardiac ring, and not at its outer edges, where the pacemaker cells that keep adult hearts ticking reside.
Jia and co. think that the early activity in heart cells, before the first contraction, might stimulate cardiovascular development.
And given the similarities between zebrafish and chick, rat, and mouse embryos, the researchers think the underlying mechanisms of heart formation might be shared across vertebrates – that group of backboned animals which includes us humans.
If so, the study could lead to further research looking at how cardiac irregularities such as arrhythmias arise in humans.
"By looking at how the heart develops," explains systems biologist Sean Megason, of Harvard Medical School, "we can see how different control mechanisms are layered on, which may tell us something about what happens if they break down."
The study has been published in Nature.