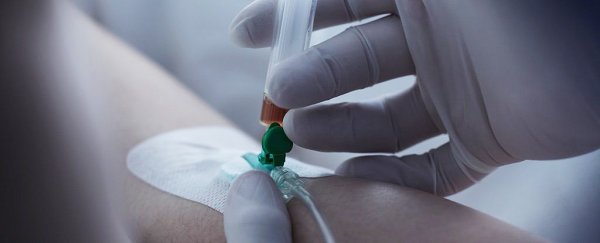
In a bold first-of-its-kind experiment, scientists have edited a person's genes directly inside living tissue in an ambitious bid to cure a man of a rare, crippling genetic disorder.
While CRISPR has broken ground in things like editing human embryos and injecting patients with genetically edited cells, this alternative technique pioneers a new real-time approach to infusing a person's blood with a gene-editing virus.
"For the first time, a patient has received a therapy intended to precisely edit the DNA of cells directly inside the body," says CEO of Sangamo Therapeutics, Sandy Macrae, whose company is testing the experimental procedure.
"We are at the start of a new frontier of genomic medicine."
The patient on the edge of that frontier is 44-year-old Brian Madeux from Arizona, who has Hunter syndrome (aka mucopolysaccharidosis II) – a serious, progressively debilitating genetic disease caused by the deficiency of an enzyme called iduronate–2-sulfatase (I2S).
“It’s kind of humbling. I’m willing to take that risk. Hopefully it will help me and other people.”
— Kevin Davies (@KevinADavies) November 15, 2017
—Hunter Syndrome patient Brian Madeux makes history as first gene editing patient in US $SGMO #notcrispr congrats @MMarchioneAP https://t.co/2B1xInsFyx pic.twitter.com/wjbH16vY1I
Due to a gene mutation, people with Hunter syndrome aren't able to properly break down large sugar molecules called glycosaminoglycans (GAGs), which build up in the body and cause a range of severe health problems, infections, and physical abnormalities that can ultimately lead to a dramatically reduced life expectancy.
In Madeux's case, the complications have resulted in 26 operations to treat things like hernias, bones pinching his spinal column, and ear and eye impairments.
"It seems like I had a surgery every other year of my life," Madeux told AP, adding that last year he nearly died from a bronchitis and pneumonia attack that warped his airway.
"I was drowning in my secretions, I couldn't cough it out."
In an effort to put a permanent stop to the gene-fuelled trauma, Madeux was connected to an IV drip for three hours on Monday by Sangamo researchers in California.

The experimental therapy, called SB-913, involves a gene-editing technique called zinc finger nucleases, which uses a modified virus engineered to precisely deliver billions of corrective genes to the patient's liver cell DNA.
Functionally similar gene therapy treatments have been around for decades, but what sets SB-913 apart from previous techniques is its precision.
"You know exactly where you're going in the genome," paediatrician Chester Whitley from the University of Minnesota – and principal investigator on Sangamo's clinical trial – told The Atlantic.
"It's not like using a shotgun hoping you're hitting a bird. It's like using a rifle."
Despite the advancements, nobody knows for sure what will happen in Madeux's brave test run, but researchers are hoping to roll out the testing on up to 30 adults to check the safety of the SB-913 procedure.
After that, the company hopes to use the therapy to treat children with Hunter syndrome and similar conditions, who would benefit from early intervention before the worst and most threatening symptoms are established.
According to the researchers, only 1 percent of Madeux's liver cells would have to be corrected to successfully treat the syndrome, with initial results expected within a month, and a fuller analysis to come in three months.
While we wait, all eyes will be on Madeux, a former chef who used to partially own two restaurants, but can no longer work due to his condition.
Even if the trial proves a complete success, SB-913 won't be able to reverse the health problems he's already incurred, but there's a chance it could prevent his situation worsening further.
The bigger goal is finding out whether this kind of gene editing can mean others won't have to go through the crippling hardships he had to.
"It's kind of humbling," Madeux told AP.
"I'm willing to take that risk. Hopefully it will help me and other people."