If you flick a handful of droplets on a very hot pan, you can watch them skitter and dance about.
These droplets, believe it or not, are actually levitating. If a surface is hot enough, the heat will vaporize the side of the droplet closest to it, creating a cushion of gas on which the rest of the droplet hovers.
This is known as the Leidenfrost effect, after German physician Johann Gottlob Leidenfrost, who documented the phenomenon in the 18th century.
Now, a team of scientists has worked out a way to lower the temperature at which this little water dance occurs. A surface with a microscopic texture transfers heat to the droplets more effectively, a finding that has implications for heat transfer applications – such as cooling industrial machinery, and nuclear cooling towers.
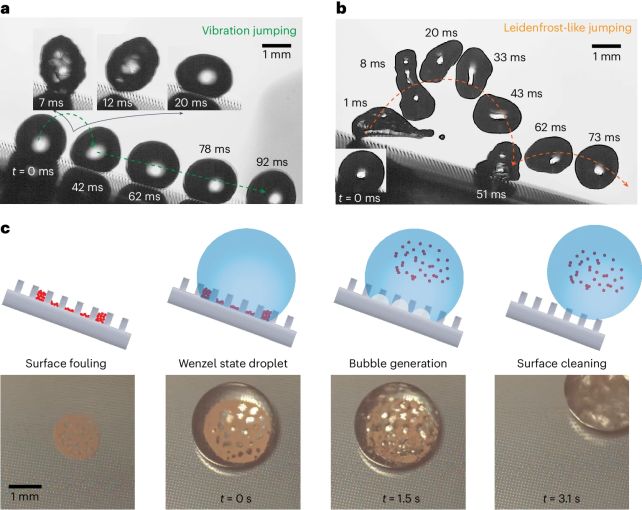
"We thought the micropillars would change the behaviors of this well-known phenomenon, but our results defied even our own imaginations," says mechanical engineer Jingtao Cheng of the Virginia Polytechnic Institute and State University.
"The observed bubble-droplet interactions are a big discovery for boiling heat transfer."

We've known about the Leidenfrost effect for some time, and its parameters are well understood. For it to occur, it needs enough heat that the water forms a vapor instantly on contact with the hot plate, but not so much heat that the entire water droplet instantly vaporizes.
The reason the water doesn't entirely vaporize at Leidenfrost temperatures is because a good proportion of the energy from the hot surface is diverted away as vapor, instead of entering the rest of the droplet.
The surface Cheng and his colleagues devised consists of hundreds of tiny little pillars about 0.08 millimeters high, about the width of a human hair. These are arranged in a grid, separated by a distance of about 0.12 millimeters. When placed on the surface, a water droplet covers about 100 of the pillars.
As the water sits on the surface, the pillars press into the water droplet, imparting more heat to the interior, and allowing the water to boil more quickly. This means that the Leidenfrost effect can be observed, within milliseconds, and at much lower temperatures than it can with a flat surface like a hotplate or frying pan.
In fact, the team was able to induce Leidenfrost levitation at 130 degrees Celsius, far lower than the 230 degrees Celsius they estimated as typical for the effect under these conditions.
Now, water is an excellent medium for cooling. Water boils and vaporizes at around 100 degrees Celsius (it varies a little by altitude). Liquid water cannot be hotter than this boiling point, since it turns into vapor.
That's why this person was able to cook soup in a plastic bag over a fire: the heat is transferred to the water, which cannot exceed the melting point of the plastic (note: do not do this, there are chemicals in plastic that you don't want in your soup).
The micropillar surface therefore offers a more efficient heat transfer mechanism that could be a lot safer than water cooling technologies currently in use, the researchers say, helping to prevent dangerous accidents such as vapor explosions.
"Vapor explosions occur when vapor bubbles within a liquid rapidly expand due to the [presence of an] intense heat source nearby. One example of where this risk is particularly pertinent is in nuclear plants, where the surface structure of heat exchangers can influence vapor bubble growth and potentially trigger such explosions," says engineer Weng Huang of Virginia Tech.
"Through our theoretical exploration in the paper, we investigate how surface structure affects the growth mode of vapor bubbles, providing valuable insights into controlling and mitigating the risk of vapor explosions."
The team's research has been published in Nature Physics.
