As infectious bacteria become increasingly resistant to antibiotics, researchers are looking for new ways to combat the so-called 'superbugs' that are predicted to kill 300 million people by 2050 if we don't find a solution.
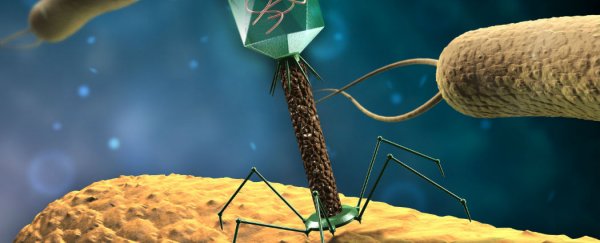
And now a team of microbiologists at Tel Aviv University in Israel has devised a way for bacteria-killing viruses - known as phages - to make superbugs become sensitive to our antibiotics once more.
The researchers described their proof-of-concept in Proceedings of the National Academy of Sciences last week. It involved developing two types of phages that target the common gut bacterium, E. coli.
One was a 'lytic' phage that killed the bacterium. But researchers also used a method for gene-editing called CRISPR-Cas in a 'temperate' phage that could then inject the E. coli with specifically prepared DNA. This hijacking both destroyed the E. coli's antibiotic resistance, and protected them from the murderous lytic phage.
Attacked by deadly phages on the one hand, and DNA-hijacking phages on the other, the bacteria in this experiment were forced to change their path of evolution. A bacterium that can't be killed by the lytic phage has an advantage over a bacterium that can be. As a result, the hijacked E. coli, even though they were sensitive to antibiotics, survived the phage attack and took over the population.
As the team noted in their paper, "[s]ensitising bacteria to various antibiotic resistance genes may easily be accomplished by the CRISPR-Cas system, as it can be programmed to eliminate any gene of interest."
The method "demonstrates that bacteria can be sensitised to approved and useful antibiotics following delivery of a rationally designed CRISPR-Cas system," the researchers report, adding that, "It does not aim to directly kill treated bacteria, but rather to sensitise them to antibiotics and to kill the non-sensitised bacteria."
Most phages only attack one species of bacteria, which means that it's possible to use this method to only target one bacterial population without disrupting the others. However, the researchers say that most pathogens do have a corresponding virus somewhere out there that would be happy to attack it. Therefore it might be possible to design a whole army of phages that can each take on a wider range of the bacterial species. It would make for an impressive, albeit invisible, war campaign.
Unlike antibiotics, which have small molecules, phages are difficult to insert into living tissue. But with this method, we could treat bacterial populations outside the body. The authors state that these designer phages could be added to hand sanitisers and surface-cleaning sprays in hospitals, where they would proceed to wage war on the superbugs. After the phage assault, more and more bacteria in the population would remain antibiotic-sensitive, and this might even prevent new resistant bugs from emerging.
As the team concludes in the paper, if their strategy is used broadly, it would potentially change the nature of hospital-acquired infections "by making the bacteria more susceptible rather than more resistant to antibiotics".
See below for more details on how CRISPR-Cas works: