Thanks to the way chemical bonds between molecules work, phase transitions are generally pretty simple - when a substance hits its freezing point, it starts to solidify, and when it hits its boiling point, it starts to vaporise.
But scientists have figured out how to seriously mess with these basic transitions, because for the first time, they've managed to melt solid metal bismuth into a liquid at temperatures well below its freezing point.
To help you visualise that craziness, it's like freezing a bowl of water into ice by dropping the temperature to lower than 0°C (32°F), and then, without changing the temperature at all, forcing that freezing cold ice to start melting back into a liquid.
As the team from the Carnegie Institution of Washington's Geophysical Laboratory points out, we're coming to realise that there are two 'flavours' of phase transitions at play in the world, and one of them is far more random than we could have imagined.
The first 'flavour' describes the simple phase transitions we learn about in school (liquid to solid, liquid to gas), that are governed by chemical bonds that remain intact.
In this type of phase transition, the chemical bonds don't break as the material goes from one phase to another, but they do alter in orientation and length to allow for major, but reversible, physical changes.
And then there's the strange phenomenon of 'reconstructive phase transitions', which are far more chaotic and hard to predict, because they involve breaking certain parts of the chemical bonds, causing significant structural shifts in the material as it enters a new phase.
But, oddly enough, this random type of phase transition is far more prevalent in nature than the 'simple' ones.
"We experience phase transitions almost all the time," one of the team, Guoyin Shen, told Ryan F. Mandelbaum at Gizmodo, "but we actually know very little about them."
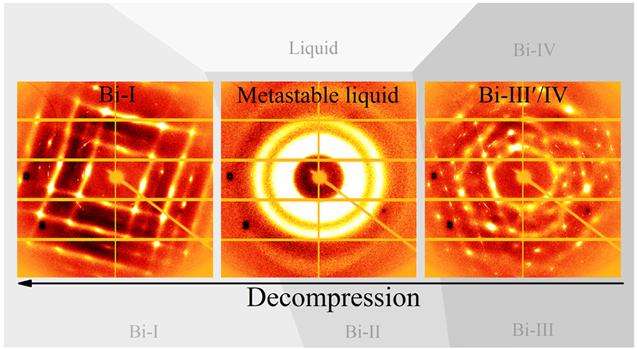
Shen and his team took a piece of crystalline bismuth (that amazing, rainbow-hued metal) in a diamond anvil cell, and subjected it to pressures and decompression ranging from 32,000 times atmospheric pressure (3.2 GPa) to 12,000 atmospheres (1.2 GPa) at a temperature of 216°C (420°F or 489 K).
That temperature is important, because bismuth's melting point is 271.52°C (520.74°F or 544.67 K), which means at temperatures below this point, the metal should remain a solid.
Once the team had subjected their bismuth to 32,000 atmospheres, they slowly started to decompress it.
At the 23,000 atmospheres, it started to melt into a liquid, and remained that way until 15,000 atmospheres.
Below 15,000 atmospheres, the metal started to recrystallise, and once it hit 12,000 atmospheres, it had transitioned back into a solid - no changes in temperature required.
 Solid bismuth at 32,000 atmospheres (right), liquified bismuth at 23,000 atmospheres (middle), and recrystallised bismuth at 12,000 atmospheres (left). Credit: Chuanlong Lin and Guoyin Shen, Carnegie Institution
Solid bismuth at 32,000 atmospheres (right), liquified bismuth at 23,000 atmospheres (middle), and recrystallised bismuth at 12,000 atmospheres (left). Credit: Chuanlong Lin and Guoyin Shen, Carnegie Institution
Referring to this unexpected liquid phase as a 'metastable liquid', the team found that it could last for hours below the melting point of bismuth, so long as the conditions remained the same.
But unlike simple phase transitions based on intact chemical bonds, if you disturb a metastable liquid, it'll quickly transition back into the phase it was 'supposed' to be in.
"Metastable liquids have found a little nook in the laws of physics where they can stay liquid-like - but like a spinning plate balancing on a stick, any perturbation, and the atoms zip back into a solid form," says Mandelbaum.
The researchers also found that this metastable liquid formed in a similar pressure-temperature range to that of supercooled bismuth - another form of reconstructive phase transition that scientists are far more familiar with.
When a material is supercooled, it means a liquid has been cooled to well below its freezing point without turning into a solid or a crystal.
Water, for example, usually freezes at 0°C (32°F or 273.15 K), but can be supercooled at certain pressures at the −48.3 °C (−55°F or 224.8 K) range, which keeps it liquid, instead of icy.
Now that we know there are two types of reconstructive phase transitions - supercooling and metastable liquids - the researchers say it's a pretty good indication that they're more common in nature than we thought.
And now that we have our first evidence that this phenomenon can actually occur, we know what to look for in other strange substances - on Earth and beyond - and it could help us produce material forms we haven't even imagined yet.
"Because reconstructive phase transitions are the most fundamental type, this research provides a brand new way for understanding how different materials change," Shen said in a press release.
"It's possible that other materials could display a similar metastable liquid when they undergo reconstructive transitions, and that this phenomenon is more prevalent than we thought. The results will no doubt lead to countless surprises in both materials science and planetary science in the coming years."
The research has been published in Nature Communications.
