A fascinating parasitic plant found lurking in clusters around the bases of trees has optimized its living strategy so well it's been able to prune nearly a third of its genome.
The genus, known as Balanophoraceae, might be mistaken for a strange, fleshy, pink fungus, poking almost phallically out of the leaf litter. With a pungent aroma akin to that of carnal rot, the parasite is far from plant-like.
But what appears to be a mushroom cap is actually a flower head, its roots tightly enmeshed in those of the tree it lurks beneath – a successful parasitic relationship, where Balanophora slurps nutrients from the other organism.
Led by geneticist Xiaoli Chen of BGI Research in China, a team of scientists discovered rather than extending a growth known as a haustorium into its host to steal nutrients as other plant parasites do, Balanophora mingles its flesh with its host's vascular system underground in a tuber-like organ. This organ is a chimeric structure, where host and parasite genetic material commingles.
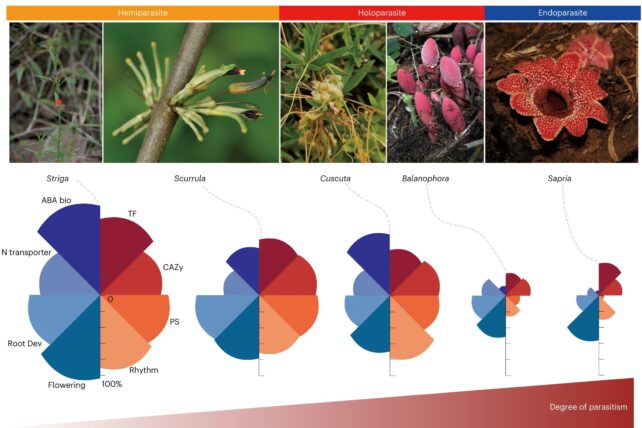
In addition, Balanophora's genome reduction – extreme even for a parasite – closely resembles that of Sapria: another, but very different, genus of parasitic plant. This is a fascinating example of convergent evolution, whereby very different organisms separately develop very similar traits over time.
"The extent of similar, but independent gene losses observed in Balanophora and Sapria is striking," Chen says. "It points to a very strong convergence in the genetic evolution of holoparasitic lineages, despite their outwardly distinct life histories and appearances, and despite their having evolved from different groups of photosynthetic plants."
Parasites are marvelous things, really. Their survival strategy is to find other species that are doing well, and reap the benefits for themselves, in various ways. This can sometimes benefit the host, too; and it's a fantastic driver of evolution.
But when you're sapping life from another organism, you might not necessarily need all the biological functions you started with before turning to a life of crime. Many plant parasites have evolved to become highly dependent upon their hosts, and their genomes have shrunk accordingly.
Parasites like Balanophora and Sapria are known as obligate or holoparasites, and are utterly dependent on their host. Both have even lost the ability to photosynthesize, which is how green plants obtain their nutrients.
Chen wanted to know how these plants came to reach their current form, and decided to take a closer look at their genomes.
This quickly got cool… and weird. Sapria is a pretty extreme example of a holoparasite. It's lost, over time, 38 percent of its genome. But Balanophora isn't far behind. It has lost a rather striking 28 percent of its genome.
Even more strikingly, both parasites have lost a similar set of genes. Obtaining nutrients from a host means that the genes involved in photosynthesis are no longer needed. The two parasites have also retained genes related to metabolism; they still need some means of processing the stolen nutrients.
Interestingly, the researchers found both plants have lost the genes for synthesizing abscisic acid, a hormone used by plants for signaling and stress responses. But while Balonophora can't make abscisic acid, it can still use it for signaling. This suggests that it may have lost genes that would overlap with some host functions, so as not to harm the host.

"The majority of the lost genes in Balanophora are probably related to functions essential in green plants, which have become functionally unnecessary in the parasites," says botanist Sean Graham of the University of British Columbia.
"That said, there are probably instances where the gene loss was actually beneficial, rather than reflecting a simply loss of function. The loss of their entire abscisic acid biosynthesis pathway may be a good example. It may help them to maintain physiological synchronization with the host plants. This needs to be tested in the future."
This means some gene loss in parasites isn't just a loss of unnecessary function, but an adaptation to the parasite lifestyle. It's a fascinating revelation that could help us better understand how parasitic and symbiotic relationships evolve between organisms.
The team suggests that other holoparasitic plants might display similar traits that could help not just study parasite-host relationships, but how plants communicate with each other.
"The study of parasitic plants deepens our understanding of dramatic genomic alterations and the complex interactions between parasitic plants and their hosts," says geneticist Huan Liu of BGI Research.
"The genomic data provides valuable insights into the evolution and genetic mechanisms behind the dependency of parasitic plants on their hosts, and how they manipulate host plants to survive."
The research has been published in Nature Plants.
