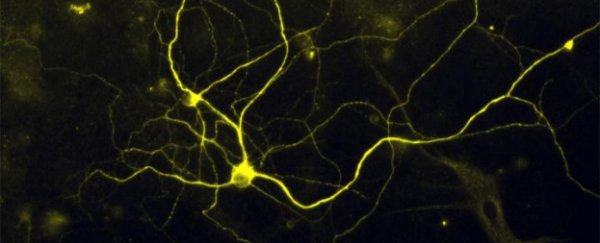
After six years of failed attempts, scientists have managed to convert mouse and human skin cells into the nerve cells that transmit painful stimuli to our brain - groundbreaking research that will allow them to study how individuals react to different kinds of pain and develop better pain medication.
It's the first time anyone has ever been able to create fully mature, pain-sensing human nerve cells in the lab, and it's pretty incredible, as the team from Harvard University in the US managed to do it using ordinary skin cells, rather than stem cells.
The model, which they're calling "pain in a dish", could be the key to understanding pain, which remains one of the most mysterious yet crucial of human sensations.
The adult mouse and human skin cells were converted into neurons using a novel combination of five transcription factors, and they can respond to both the intense, "ouch" pain triggered by an injury, and the more subtle, itchy pain caused by inflammation.
This dual ability suggests that the neurons are functioning in much the same way that they do in our bodies, according to nervous system specialist Clifford Woolf from the Harvard Stem Cell Institute, who led the research.
The team has already tested the neurons' response to several stimuli, and shown that they start to fire off signals when they're heated above 42 degrees Celsius, or are put into contact with capsaicin, the active component of chilli peppers. The results are published in Nature Neuroscience.
"Pain is arguably one of the most important of our sensory applications," Woolf told Steve Connor from the Independent. "It warns us of danger in the environment and we're exposed to a lot of things that can damage our sensitive biological systems."
"The goal was to study the key aspects of how the nervous system generates the sense of pain. The idea was to create the key elements of the human nervous system involved in pain in a dish," he added.
Woolf's team have been working to achieve this goal for the past six years, but were originally trying to create these neurons using embryonic stem cells.
"We spent three years trying to recapitulate the developmental steps involved, and it turned out to be a total bust," said Woolf in a press release.
Eventually in desperation they started looking at turning mouse and human skin cells directly into neurons, and found that they were surprisingly simple to convert.
"We took mature pain neurons from mice, and found transcription factors that hadn't been described in them before," said Woolf. They then used those factors, along with two previously known factors, to transform them the adults cells into the neurons that control the pain we feel.
Currently, there are hundreds of thousands of people around the world living with chronic pain and itching, and it's something that scientists still don't understand very well.
But this new research will allow us to create disease-specific pain models in the lab and then work out what causes specific kinds of pain, and how it can be treated.
"I think the ability to make human pain neurons for the pain field is going to be very important," Woolf said in the press release.
"Furthermore, our failure with embryonic stem cells lead us to work with adult tissue samples making the technology much more clinically relevant since these are easy to collect from patients suffering from different kinds of pain."
Source: ScienceDaily, The Independent