Researchers have discovered that a particular protein in the gut is fighting an important battle against the spread of bacteria.
Through a combination of protective effects, the protein could in the future be used in treatments for conditions such as inflammatory bowel disease.
The protein, intelectin-2, isn't new to scientists, but its role in the gastrointestinal (GI) tract hasn't been clear. Intelectin-2 is part of a group of proteins called lectins, which work by binding to specific sugar molecules.
A team led by researchers at MIT has now found that intelectin-2 works in two ways: First, it links mucus molecules lining the GI tract, strengthening the mucus barrier that protects intestinal tissues.
But if there's a breach, intelectin-2 also recognizes and traps a variety of bacterial cells, either inhibiting their growth or killing them off completely.
So intelectin-2's role is a combination of defense and offense activities that protect bodily health.
"What's remarkable is that intelectin-2 operates in two complementary ways," says MIT chemist Laura Kiessling.
"It helps stabilize the mucus layer, and if that barrier is compromised, it can directly neutralize or restrain bacteria that begin to escape."
The researchers used a combination of techniques to look at the activity of intelectin-2. They studied mouse tissue, gut bacteria from mice and humans, and both the human and mouse versions of the protein isolated in the lab.
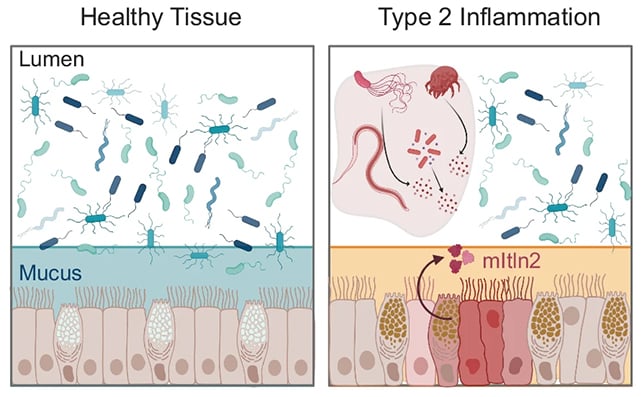
These approaches revealed that intelectin-2 targets galactose, a simple sugar found in mucus molecules and in the carbohydrates that cover the surface of some bacterial cells (including bacteria linked to GI infections).
In scenarios where intelectin-2 binds to and traps microbes, the bacteria seem to break down over time, possibly because the protein disrupts their outer membrane.
What's more, some of the bacteria that intelectin-2 neutralizes have developed antibiotic resistance to varying degrees, making these discoveries even more valuable in terms of drug development.
Those bacteria include Staphylococcus aureus, which can trigger sepsis, and Klebsiella pneumoniae, which causes pneumonia and other infections.
"Intelectin-2 first reinforces the mucus barrier itself, and then if that barrier is breached, it can control the bacteria and restrict their growth," says Kiessling.
Scientists estimate that more than 200 lectins are encoded in the human genome, so the work continues to understand how they influence cell-to-cell communication and immune function.
Before this study of intelectin-2, researchers had previously linked intelectin-1 in the gut to Crohn's disease. So it seems these proteins have crucial parts to play in protecting gut health.
The researchers also point out that intelectin-2 levels are often found at unusually low or high levels in people with inflammatory bowel disease. Based on this new study, that may indicate the mucus barrier isn't being patched up well enough, or that healthy bacteria are being killed off.
Related: Microbe in Human Gut May Boost Muscle Strength, Study Finds
Both the offense and defense aspects of intelectin-2's operations might be useful in the future, either through the development of drugs that mimic some of the actions the protein takes, or by boosting the protein's own capabilities in the body.
These lectins are ancient animal proteins, believed to predate the more advanced immune systems working in our bodies today. But this research shows they're not blunt instruments; they're very precise in taking down dangerous bacteria.
"Harnessing human lectins as tools to combat antimicrobial resistance opens up a fundamentally new strategy that draws on our own innate immune defenses," says Kiessling.
"Taking advantage of proteins that the body already uses to protect itself against pathogens is compelling and a direction that we are pursuing."
The research has been published in Nature Communications.