The origins of Alzheimer's remain contentious, but a new study suggests the disease may emerge as two key proteins compete inside brain cells.
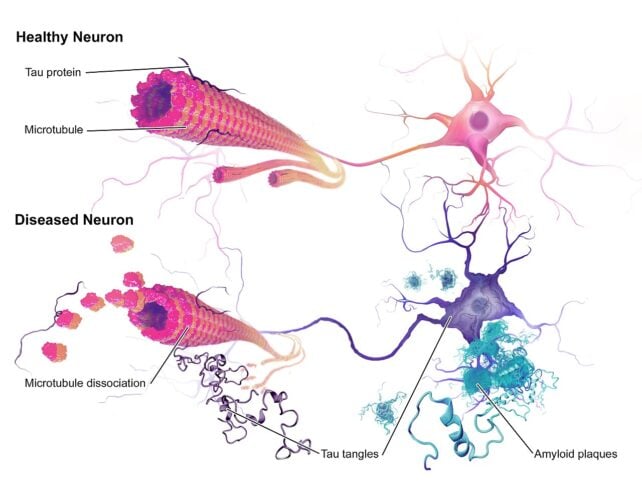
Alzheimer's disease, the most common form of dementia, has long been associated with the build-up of two proteins in the brain: amyloid-beta and tau.
This new study ties those two together, offering a "unifying theory" that, according to the team of chemists proposing it, resolves some conflicting ideas about Alzheimer's.
Amyloid-beta peptides are sticky fragments of a larger protein that clump together to form plaques in the brains of people with Alzheimer's. These plaques, it's thought, can emerge some 20 years before symptoms appear.
But some research suggests that tau tangles, knots of misfolded tau proteins that form inside diseased neurons, are a better indicator of cognitive status in Alzheimer's than amyloid-beta plaques.
Normally, tau stablizes microtubules, the internal scaffold of cells, but in Alzheimer's disease, tau detaches from microtubules, clogging up cells.
These two hallmarks of Alzheimer's disease have been called into question in recent years, but they remain a major focus of research – with neuroscientists trying to figure out which forms first, what effect they each have, and if they truly do cause the disease or are just a symptom of it.
"In addition to having dementia, [an] Alzheimer's diagnosis requires both [amyloid-beta] and tau buildup in the brain," explains Ryan Julian, a chemistry professor at the University of California, Riverside, and the study's senior author.
"But many labs focus on the role of one and ignore the other."
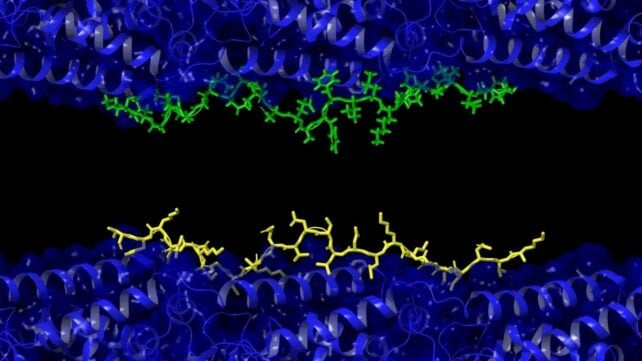
To investigate, Julian and his colleagues conducted a series of protein binding studies, focusing on how amyloid-beta and tau interact around microtubules.
Noticing that amyloid-beta peptides resembled, in sequence, the part of tau proteins that attach to microtubules, the team mixed the two proteins together in solution, along with tubulin, the building block of microtubules.
"Our work shows amyloid beta and tau compete for the same binding sites on microtubules, and that [amyloid-beta] can prevent tau from functioning correctly," says Julian.
Using fluorescently labeled amyloid-beta, the researchers could see when amyoid-beta peptides 'stole' the binding sites normally used by tau proteins.
The researchers also 'tempted' amyloid-beta with another common protein, myoglobin, and found that amyloid-beta peptides still preferred binding to microtubules – meaning they weren't just binding to any protein in solution.
Based on their results, the researchers hypothesise that they may have found a solution to the chicken-or-egg scenario between amyloid-beta peptides and tau tangles – although we should bear in mind that their experiments were with mixtures of purified proteins. Understanding how proteins behave inside cells is far more complicated.
If amyloid-beta peptides displace tau from its usual binding sites, as these protein studies suggest, then this might explain how tau can knot itself in tangles and microtubules become destablized, disrupting core functions of neurons and ultimately leading to cell death.
"The key distinction here is the recognition that tau does not initiate pathology on its own but becomes problematic after displacement by [amyloid-beta]," Julian and colleagues write in their paper.
They also suggest that this displacement of tau, leading to faulty microtubules, may be the key source of toxicity for brain cells – not the accumulation of either amyloid-beta plaques or tau tangles (though they add to the cells' problems).
"This new hypothesis contextualizes many prior observations in the literature and resolves the contradictions between the conventional hypotheses of the underlying cause of Alzheimer's disease," Julian and colleagues write.
This research adds a new perspective to the results of clinical trials testing Alzheimer's therapies that have fallen short and studies that have found clearing amyloid-beta plaques doesn't appear to repair key brain functions.
In the long run, if these findings can be backed up with further studies, the work may redirect efforts to develop effective treatments for Alzheimer's disease, which makes up to 70 percent of dementia cases and has no known cure.
Interestingly, recent animal studies have suggested that lithium may have a protective effect, helping to stablize microtubules.
It follows that developing therapies to protect these tiny tubes may be a fresh approach to explore, rather than targeting protein build-up, as most existing therapies for Alzheimer's have done.
Related: Simple Lifelong Habits Can Cut Your Alzheimer's Risk by 38%, Study Finds
The study "helps make sense of many results that previously seemed unrelated," concludes Julian.
"It gives us a clearer picture of what may be going wrong inside neurons and where new treatments might start."
This research is published in the PNAS Nexus.

