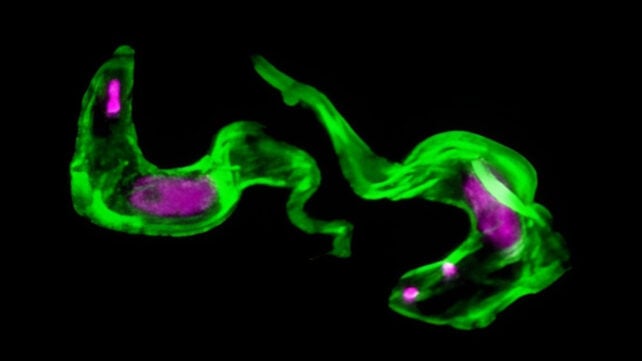
The tropical disease known as sleeping sickness is caused by the African Trypanosoma brucei parasite.
For decades, scientists have been unable to pin down exactly how this parasite successfully evades detection in its hosts – until now.
What was already known was that the bug wears a 'molecular cloak', made up of the variant surface glycoprotein (VSG) protein. This outer layer is constantly being renewed in order to evade detection by the immune system.
The mystery was the biological manufacturing mechanisms behind this constant overhaul of the coating, and in particular, why T. brucei can keep this part of its cellular machinery so active without overloading other parts of the organism.
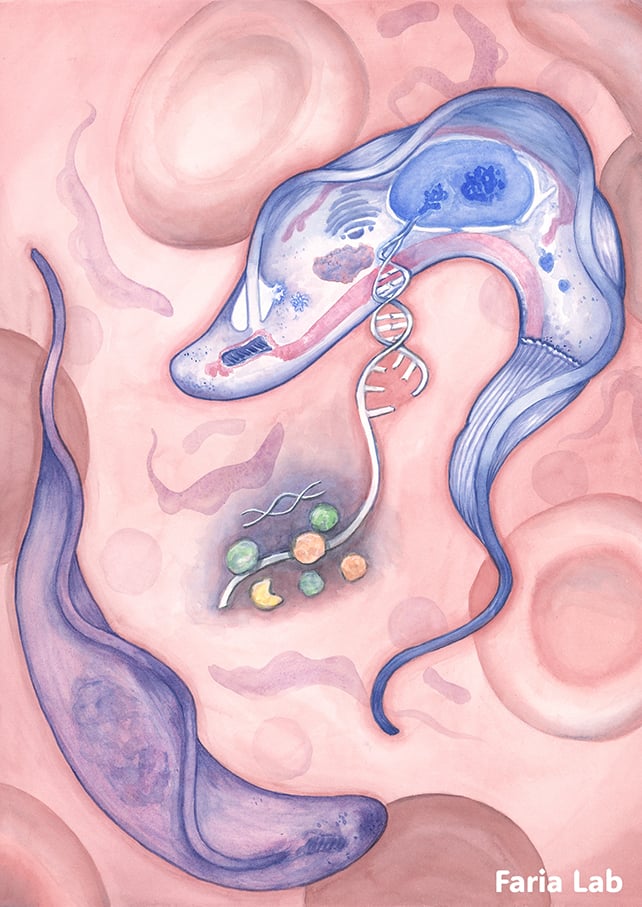
Here, a team from the University of York in the UK have found an answer: a 'molecular shredder' that the researchers named ESB2.
It lets the cloak-making instructions (in the form of RNA) pass through, while chopping up instructions for other proteins being made at the same time.
"When we first saw the molecular shredder localized in the microscope, we knew we had found something special," says microbiologist Lianne Lansink, the first author of a new study on the findings.
The parasite needs some helper proteins besides VSG in order to survive – to steal nutrients from its host, for example. However, it's the VSG protein production that's most important, and this is where ESB2 comes in.
By sitting at the end of the cell production line, ESB2 can carefully and deliberately manage the levels of each protein by intercepting RNA molecules. This means VSG production can continue at full speed, and wasted energy is minimized.
"Imagine a molecular shredder stationed right inside the genetic print room, selectively redacting specific pages of a manual as they are being printed," says microbiologist Joana Correia Faria, senior author of the study.
"It is a remarkably efficient way to downregulate genes at the source."

The researchers found ESB2 by tagging proteins inside the parasite's cells and identifying what they were through mass spectrometry. Having found ESB2, the next step was to work out what it was doing, so the researchers genetically edited T. brucei to remove it.
With ESB2 gone, production levels of the helper proteins shot up, matching the levels of VSG. The hidden role of ESB2 was revealed, and now scientists know that it's there, they can start working on treatments to target it.
"This suggests a fundamental shift in how we view infection," says Faria.
"Survival for many organisms may depend less on how they issue genetic instructions and more on how they destroy them at the source."
By disrupting the activity of ESB2, treatments might be able to destroy T. brucei at the same time, or at least make it more visible to the body's immune system. Any treatments are still a way away, but some of the parasite's trickery has now been exposed.
Sleeping sickness is transmitted by the bite of the tsetse fly, and can cause numerous issues with the central nervous system. The disease can lead to issues sleeping, feelings of confusion, and comas. It can also be fatal.
While the number of people with the disease has been reduced in recent years, it continues to affect hundreds of people annually. While treatments do exist, they're not always accessible, aren't guaranteed to work, and can come with severe side effects.
Thanks to the work of the researchers behind this new discovery, there's fresh hope for finding new ways to tackle sleeping sickness.
Next, the researchers want to take a closer look at ESB2 and how it identifies different RNA molecules, as well as investigating some of the other proteins that it interacts with and may rely on.
Related: Immune Cells Do Something Unexpected to Stop This Brain Parasite From Spreading
"This discovery is a real full-circle moment for me," says Faria. "The mystery of how this parasite manages the asymmetric expression of its genetic manual has been a cold case in the back of my mind since my days as a postdoc.
"It's a testament to what a fresh lab and a diverse group of scientists can achieve when they look at an old problem from a completely new angle."
The research has been published in Nature Microbiology.